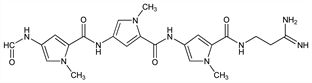
8772. Distamycin A
Nomenclature
Description and references
Antiviral antibiotic obtained from Streptomyces distallicus. Binds to the minor groove of DNA, preferentially to adenine-thymine rich sequences; inhibits DNA-dependent RNA synthesis. Isoln: F. Arcamone et al., DE 1039198 (1958 to Farmaceutici Italia), C.A. 55, 2012f (1961). Structure and synthesis: idem et al., Nature 203, 1064 (1964); eidem, Gazz. Chim. Ital. 97, 1097, 1110 (1967). Total synthesis: M. Bialer et al., Tetrahedron 1978, 2389; L. Grehn, U. Ragnarsson, J. Org. Chem. 46, 3492 (1981). Toxicity and antitumor activity: A. DiMarco et al., Cancer Chemother. Rep. 18, 15 (1962). Review: F. E. Hahn in Antibiotics vol. 3, J. W. Corcoran, F. E. Hahn, Eds. (Springer-Verlag, New York, 1975) pp 79-100. Review of mechanism of action: H. Grunicke et al., Rev. Physiol. Biochem. Pharmacol. 75, 69-96 (1976). Review of use for targeted delivery of antitumor agents: P. G. Baraldi et al., Bioorg. Med. Chem. 15, 17-35 (2007).