8803. Stavudine
Nomenclature
Description and references
Analog of thymidine, q.v.; reverse transcriptase inhibitor. Prepn: J. P. Horwitz et al., J. Org. Chem. 31, 205 (1966); J. W. Beach et al., ibid. 57, 3887 (1992). Large scale production: J. E. Starrett et al., EP 334368; eidem, US 5130421 (1989, 1992 both to Bristol-Myers). In vitro activity against HIV and toxicology: M. M. Mansuri et al., Antimicrob. Agents Chemother. 34, 637 (1990). Mechanism of action study: H.-T. Ho, M. J. M. Hitchcock, ibid. 33, 844 (1989). Disposition and metabolism: E. M. Cretton et al., ibid. 37, 1816 (1993). Clinical pharmacokinetics: M. N. Dudley et al., J. Infect. Dis. 166, 480 (1992). HPLC determn in plasma and urine: J. S. Janiszewski et al., J. Chromatogr. 577, 151 (1992). Clinical evaluation in AIDS and ARC: M. J. Browne et al., J. Infect. Dis. 167, 21 (1993).
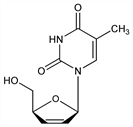
Properties
Colorless, granular solid from ethanol/benzene, mp 165-166° (Horwitz). Also reported as crystals from ethanol-ether, mp 174° (Beach). uv max (water): 266 nm (ε 10149). [α]D25 39.4° (c = 0.701 in water); [α]D20 46.1° (c = 0.7 in water).Therapeutic Category
Antiretroviral.
Keywords
Reverse Transcriptase Inhibitor; Antiretroviral; Reverse Transcriptase Inhibitors; Nucleosides/Nucleotides