9397. Thymidine
Nomenclature
CAS number: 50-89-5
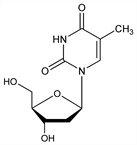
1-(2-Deoxy-β-d-ribofuranosyl)-5-methyluracil; thymine-2-desoxyriboside. C10H14N2O5; mol wt 242.23.
C 49.58%, H 5.83%, N 11.56%, O 33.03%.
Description and references
Constituent of deoxyribonucleic acid, q.v. Isoln from thymonucleic acid: Levene, London, J. Biol. Chem. 83, 793 (1929). Structure: Levene, Tipson, ibid. 109, 623 (1935). Conformation: Lemieux, Can. J. Chem. 39, 116 (1961); Tollin et al., Nature 217, 1148 (1968). Prepn of thymidine-3′-phosphate and of thymidine-5′-phosphate: Tener, J. Am. Chem. Soc. 83, 165 (1961). Review: Basic Principles in Nucleic Acid Chemistry vol. 1, P. O. P. Ts'o, Ed. (Academic Press, New York, 1974) passim.
Properties
Rosettes of needles from ethyl acetate. mp 185°. Yields a sublimate of thymine when heated. [α]D25 +30.6° (c = 1.029). uv max (pH 7.2): 206.5, 267 nm (ε × 10+3 9.8, 9.7). Absorption spectra: D. Voet et al., Biopolymers 1, 193 (1963). Sol in water, methanol, hot alcohol, hot acetone, hot ethyl acetate, pyridine, glacial acetic acid; sparingly sol in hot chloroform.Derivative
Monotrityl thymidine.C29H28N2O5; mol wt 484.54.
C 71.88%, H 5.82%, N 5.78%, O 16.51%.