8808. Stenbolone
Nomenclature
CAS number: 5197-58-0
(5α,17β)-17-Hydroxy-2-methylandrost-1-en-3-one; 2-methyl-5α-androst-1-en-17β-ol-3-one; 2-methyl-17β-hydroxy-5α-androst-1-en-3-one; stenobolone (rescinded USAN). C20H30O2; mol wt 302.45.
C 79.42%, H 10.00%, O 10.58%.
Description and references
Preparation of free alcohol and acetate: R. Mauli et al., J. Am. Chem. Soc. 82, 5494 (1960); Kaspar et al., DE 1096356 (1961 to Schering AG), C.A. 55, 27440b (1961); R. E. Counsell et al., J. Org. Chem. 27, 248 (1962); GB 925849 (1963 to Syntex).
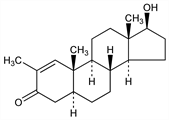
Properties
Crystals from acetone + hexane, mp 155-158°. [α]D +52° (chloroform) (Mauli). [α]D26 +47° (chloroform) (Counsell). uv max (95% ethanol): 241 nm (log ε 3.99).Derivative
Acetate.Nomenclature
CAS number: 1242-56-4
C22H32O3; mol wt 344.49.
C 76.70%, H 9.36%, O 13.93%.