8813. Stibophen
Nomenclature
CAS number: 15489-16-4
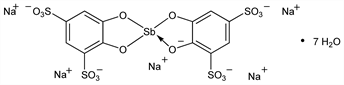
(T-4)-Bis[4,5-dihydroxy-1,3-benzenedisulfonato(4)-O4,O5]-antimonate(5) pentasodium
heptahydrate; sodium antimony bis(pyrocatechol-2,4-disulfonate) heptahydrate; antimony pyrocatechol sodium disulfonate heptahydrate; Sdt-91; Fuadin (Winthrop); Fouadin; Fantorin (Glaxo); Neoantimosan; Repodral (Winthrop). C12H4Na5O16S4Sb.7H2O; mol wt 895.23.
C 16.10%, H 2.03%, Na 12.84%, O 41.11%, S 14.33%, Sb 13.60%.
Description and references
Prepn of potassium salt: GB 213285 (1923 to Heyden). Prepn from sodium pyrocatechol-3,5-disulfonate and antimony trioxide in alkaline soln: H. Schmidt, DE 448800 (1924 to I. G. Farbenind.), Frdl. 16, 2546 (1931); idem, US 1549154 (1925); idem, Z. Angew. Chem. 43, 963 (1930); idem, US 1873668 (1932 to Winthrop). Toxicity: H. Eagle et al., J. Pharmacol. Exp. Ther. 89, 196 (1947). Clinical trial in schistosomiasis: J. Azar et al., Am. J. Trop. Med. 29, 595 (1949); in leishmaniasis: H. S. Girgla et al., Br. J. Dermatol. 97, 307 (1977). UV spectrophotometric analysis: A. Besada et al., Anal. Lett. 21, 447 (1988).
Properties
Fine crystals. Almost insol in abs alcohol, ether, chloroform, acetone, petr ether. Readily sol in cold water. Unless the colorless neutral water soln is acidified it acquires a yellowish tint and finally reaches lemon-yellow color. LD50 i.v. in rabbits: ≈90 mg/kg (Eagle).Derivative
Potassium salt.Nomenclature
Antimosan; Heyden 611.Therapeutic Category
Anthelmintic (Schistosoma).
Therapeutic Category (Veterinary)
Has been used in schistosomiasis.Keywords
Anthelmintic (Schistosoma)