8822. Streptidine
Nomenclature
CAS number: 85-17-6
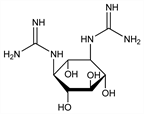
N,N′-Bis(aminoiminomethyl)streptamine; N,N′-diamidinostreptamine; N,N-(2,4,5,6-tetrahydroxy-1,3-cyclohexanediyl)bisguanidine; 1,3-diguanido-2,4,5,6-cyclohexanetetrol. C8H18N6O4; mol wt 262.27.
C 36.64%, H 6.92%, N 32.04%, O 24.40%.
Description and references
The aglycone component of the streptomycin molecule joined with streptobiosamine through a glycosidic linkage. Obtained from streptomycin by acid hydrolysis: Peck et al., J. Am. Chem. Soc. 68, 29 (1946); Fried et al., J. Biol. Chem. 162, 391 (1946). Structure: Carter et al., Science 103, 53, 540 (1946). Synthesis: Wolfrom et al., J. Am. Chem. Soc. 72, 1724 (1950). Absolute configuration: Dyer, Todd, ibid. 85, 3896 (1963); Tatsuoka, Horii, J. Antibiot. 17A, 88 (1964). Biosynthesis studies: Bruton et al., J. Biol. Chem. 242, 813 (1967).
Properties
Optically inactive, diacidic base.Derivative
Dipicrate dihydrate.C20H24N12O18.2H2O; mol wt 756.50.
C 31.75%, H 3.73%, N 22.22%, O 42.30%.
Properties
Yellow needles from water, dec 283-284° (becomes anhydrous in vacuo at 56°, dec 284-285°).Derivative
Sulfate.C8H18N6O4.H2SO4; mol wt 360.34.
C 26.67%, H 5.59%, N 23.32%, O 35.52%, S 8.90%.
Properties
Crystals from dilute sulfuric acid + acetone, not melted at 300°. Forms solvated crystals from methanol.Derivative
Dihydrochloride.C8H18N6O4.2HCl; mol wt 335.19.
C 28.67%, H 6.01%, N 25.07%, O 19.09%, Cl 21.15%.