8826. Streptomycin
Nomenclature
Description and references
Antibiotic substance produced by the soil Actinomycete Streptomyces griseus (Krainsky) Waksman et Henrici (Fam. Actinomycetaceae). Isolation: Schatz et al., Proc. Soc. Exp. Biol. Med. 55, 66 (1944). Production by aerobic fermentation and purification: Tishler in Streptomycin, Selman A. Waksman, Ed. (Williams & Wilkins, Baltimore, 1949) pp 32-54. Isoln and purification by ion exchange: Bartels et al., Chem. Eng. Prog. 54 (8), 49-51 (Aug. 1958); Bartels et al., US 2868779 (1959 to Olin Mathieson). Structure: Brink, Folkers, J. Am. Chem. Soc. 69, 1234 (1947); Wolfrom et al., ibid. 76, 3675 (1950). Total synthesis: Umezawa et al., J. Antibiot. 27, 997 (1974). Mechanism of action: B. J. Wallace et al., in Antibiotics vol. 5(pt. 1), F. E. Hahn, Ed. (Springer-Verlag, New York, 1979) pp 272-303. Review: Lemieux, Wolfrom, “The Chemistry of Streptomycin” in W. W. Pigman, M. L. Wolfrom, Adv. Carbohydr. Chem. 3, 337-384 (1948). Comprehensive description: J. Mossa et al., Anal. Profiles Drug Subs. 16, 507-609 (1986).
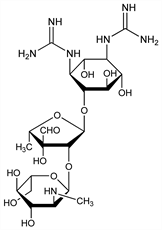
Properties
Streptomycin is usually available as the trihydrochloride, trihydrochloride-calcium chloride double salt, phosphate, or sesquisulfate, which occur as granules or powder. Odorless or nearly so, with a slightly bitter taste. Most salts are hygroscopic and deliquesce on exposure to air, but are not affected by air or light. The salts are very sol in water; but almost insol in alc, chloroform, ether. Solns are levorotatory.Derivative
Trihydrochloride.Nomenclature
Streptomycin hydrochloride.Properties
[α]D25 84°. Solubilities as determined by Weiss et al., Antibiot. Chemother. 7, 374 (1957) in mg/ml at about 28°: water >20; methanol >20; ethanol 0.90; isopropanol 0.12; isoamyl alcohol 0.117; petr ether 0.02; carbon tetrachloride 0.042; ether 0.01.Derivative
Trihydrochloride-calcium chloride double salt.Nomenclature
Streptomycin hydrochloride-calcium chloride complex.Description and references
Prepn from the trihydrochloride: Peck, US 2446102 (1948 to Merck & Co.).
Properties
Very hygroscopic, dec about 200°. [α]D25 76°.Derivative
Pantothenate.Nomenclature
Streptothenat (Grünenthal).Description and references
Prepn: GB 771338 (1957 to Grünenthal). The commercial prepn may contain the sulfate.
Derivative
Sesquisulfate.Nomenclature
Properties
White to light gray or pale buff powder with faint amine-like odor. Solubilities as determined by Weiss et al., loc. cit., in mg/ml at about 28°: water >20; methanol 0.85; ethanol 0.30; isopropanol 0.01; petr ether 0.015; carbon tetrachloride 0.035; ether 0.035.Therapeutic Category
Antibacterial (tuberculostatic).
Therapeutic Category (Veterinary)
Antibacterial.Keywords
Antibacterial (Antibiotics); Aminoglycosides; Antibacterial (Tuberculostatic)