Nomenclature
CAS number: 57-24-9
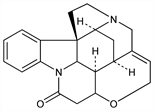
Strychnidin-10-one.
C
21H
22N
2O
2; mol wt 334.41.
C 75.42%, H 6.63%, N 8.38%, O 9.57%.
Description and references
Occurs most abundantly in seeds of Strychnos
nux-vomica L., Loganiaceae and beans of S. ignatti, Berg. One of the first alkaloids isolated in pure form. Isoln:
Pelletier, Caventou, Ann. Chim. Phys. 8, 323 (1818); ibid. 10, 142 (1819).
Extraction procedure: C. Srinivasulu et al., Res. Ind. 23, 224 (1978). Structure
elucidation: L. H. Briggs et al., J. Chem. Soc. 1946, 903; H. T. Openshaw, R.
Robinson, Nature 157, 438 (1946); R. B. Woodward et al., J. Am. Chem. Soc. 69, 2250 (1947). Total
synthesis: R. B. Woodward et al., ibid. 76, 4749 (1954); eidem, Tetrahedron 19, 247 (1963). Abs config:
A. F. Peerdeman, Acta Crystallogr. 9, 824 (1956); K. Nagarajan et al., Helv. Chim. Acta 46, 1212 (1963).
Stereoselective total synthesis: S. D. Knight et al., J. Am. Chem. Soc. 115, 9293 (1993). 1H- and 13C-NMR analysis: E.
Wenkert et al., J. Org. Chem. 43, 1099 (1978). Pharmacology and pharmacokinetics:
S. Weiss, R. A. Hatcher, J. Pharmacol. Exp.
Ther. 19, 419 (1921). Acute toxicity: I.
Setnikar, M. J. Magistretti, Arzneim.-Forsch. 14, 996 (1964). HPLC determn in urine and tissues:
T. Egloff et al., J. Clin. Chem.
Clin. Biochem. 20, 203 (1982); GC/EI-MS determn
in whole blood: M. Barroso et al., J. Chromatogr. B 816, 29 (2005). Review: J. B. Hendrickson in The Alkaloids vol. VI, R. H. F. Manske, Ed. (Academic Press, New York,
1960) pp 179-195; G. F. Smith, ibid. vol. VIII (1965) pp 591-671. Comprehensive description: F. J. Muhtadi, M.
S. Hifnawy, Anal. Profiles Drug Subs. 15, 563-646 (1986). Brief review of synthesis: U. Beifuss, Angew. Chem. Int. Ed. 33, 1144-1149
(1994).
Properties
Brilliant, colorless cubes from chloroform-ether, mp 275-285°. d18 1.359. [α]D18 104.3° (c = 0.254 in alc); [α]D25 139° (c = 0.4 in chloroform). pKa (25°) 8.26:
A. J. Everett et al., J. Chem.
Soc. 1957, 1120. uv max (95% ethanol): 255, 280, 290 nm (E1%1 cm 377, 130,
101): A. I. Biggs, J. Pharm. Pharmacol. 13, 547 (1952). One gram dissolves in 182
ml ethanol, 6.5 ml chloroform, 150 ml benzene, 250 ml methanol, 83
ml pyridine; very slightly sol in ether, water. LD50 i.v. (slow infusion) in rats:
0.96 mg/kg (Setnikar,
Magistretti).Derivative
Hydrochloride dihydrate.
Nomenclature
CAS number: 6101-04-8
C
21H
22N
2O
2.HCl.2H
2O; mol wt 406.90.
C 61.99%, H 6.69%, N 6.88%, O 15.73%, Cl 8.71%.
Properties
Efflorescent, trimetric prisms. One gram dissolves
in ≈35 ml cold water, ≈80 ml alc. Insol in ether. pH of 0.01M soln 5.4.Derivative
Nitrate.
Nomenclature
CAS number: 66-32-0
C21H22N2O2.HNO3.
Properties
Colorless, odorless needles or white, cryst powder.
At 25°, one gram dissolves in 42 ml water, 120 ml alcohol, 156 ml
chloroform. Insol in ether. pH ≈5.7. Protect
from light.Derivative
Sulfate.
Nomenclature
CAS number: 60-41-3
(C21H22N2O2)2.H2SO4.
Properties
Usually crystallizes as pentahydrate. Properties
of pentahydrate: colorless, odorless, very bitter crystals, or white,
cryst powder. Effloresces in dry air; loses all its water of crystn
at 100°. mp when anhydr ≈200° with decompn. At 25°, one gram dissolves in 31 ml water, 65
ml alcohol, 325 ml chloroform. Insol in ether. pH 5.5 in 1:100 soln.
Protect from light. LD50 orally in rats: 5 mg/kg, E. W. Schafer, Toxicol. Appl. Pharmacol. 21, 315
(1972).Caution
Extremely poisonous, see: G. D. Osweiler, Curr. Vet. Ther. 6, 115 (1977). Potential symptoms of overexposure to
strychnine are stiff neck and facial muscles; restlessness, apprehension
and increased acuity of perception; increased reflex excitability;
cyanosis; tetanic convulsions with opisthotonos. See NIOSH Pocket Guide to Chemical Hazards (DHHS/NIOSH
97-140, 1997) p 286.Use
Chiefly in poison baits for rodents.
Therapeutic Category (Veterinary)
Has been used as a tonic and central
stimulant.
