8879. Sucralfate
Nomenclature
Description and references
A basic aluminum sucrose sulfate complex which inhibits peptic hydrolysis and stomach acidity. Prepn: M. Nametaka et al., Yakugaku Zasshi 87, 889 (1967), C.A. 68, 20831d (1968); FR 1500571; N. Yoshihiro et al., US 3432489 (1967, 1969 both to Chugai). Structure and properties: R. Nagashima, N. Yoshida, Arzneim.-Forsch. 29, 1668 (1979). In vitro and in vivo study of antipeptic and antiulcer activity: L. E. Borella, et al., ibid. 793. Series of articles on mode of action: ibid. 30, 73-88 (1980). Pharmacokinetics, metabolism and selective binding studies: K. Steiner et al., ibid. 32, 512 (1982). Clinical studies: J. F. Mayberry et al., Br. J. Clin. Pract. 32, 291 (1978). Symposium on clinical studies: J. Clin. Gastroenterol. 3, Suppl. 2, 103-184 (1981); Scand. J. Gastroenterol. 18, Suppl. 83, 1-82 (1983). Review of pharmacology and clinical studies: R. N. Brogden et al., Drugs 27, 194 (1984). Series of articles on clinical efficacy in peptic ulcer disease and gastritis: Am. J. Med. 79, Suppl. 2C, 1-64 (1985).
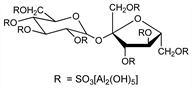
Properties
White amorphous powder. Sol in dil HCL and NaOH solns. Practically insol in water, ethanol, CHCl3. pKa = 0.43 to 1.19. Dissolution of aluminum occurs at pH <3; of sucrose sulfate at pH >4.Therapeutic Category
Antiulcerative.
Keywords
Antiulcerative; Cytoprotectant (Gastric)