Nomenclature
CAS number: 57-50-1
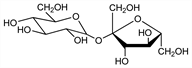
β-
d-Fructofuranosyl-α-
d-glucopyranoside; α-
d-glucopyranosyl-β-
d-fructofuranoside; sugar; saccharose; cane sugar; beet sugar.
C
12H
22O
11; mol wt 342.30.
C 42.11%, H 6.48%, O 51.41%.
Description and references
Obtained from sugar cane (Saccharum officinarum L., Gramineae) and sugar beet (Beta valgaris L., Chenopodiaceae). Sugar cane contains from 15-20%
and sugar beet from 10-17% sucrose. Structure: Avery et al., J. Chem. Soc. 1927, 2308; Beevers, Cochrane, Proc. Roy. Soc. 190A, 257 (1947). Synthesis: Pictet, Vogel, Helv. Chim. Acta 11, 436 (1928);
Lemieux, Huber, J. Am. Chem. Soc. 78, 4117 (1956). Ref. with extensive bibliography:
Bates, Polarimetry, Saccharimetry, and the
Sugars, National Bureau of Standards Circular C440, Washington,
1942; W. Pigman, The Carbohydrates (Academic Press, New York, 1957) pp 501-506. Reviews: M. R. Jenner, Dev. Food Carbohydr. 2, 91-143 (1980); R. Khan, Pure
Appl. Chem. 56, 833 (1984).
Properties
Monoclinic sphenoidal crystals, cryst masses,
blocks, or powder. Sweet taste. Stable in air. Finely divided sugar
is hygroscopic and absorbs up to 1% moisture which is given up on
heating to 90°. d425 1.587. Dec 160-186°. Chars and emits characteristic
odor of caramel. [α]D20 not
less than +65.9° (c = 26); usual
value [α]D25 +66.47 to +66.49°. One gram dissolves in 0.5 ml water;
in slightly more than 0.2 ml boiling water, in 170 ml alcohol; in
about 100 ml methanol. Moderately sol in glycerol, pyridine. pKa 12.62. d420 of water solns (g/100 g): 2% 1.0060; 6% 1.0219; 10% 1.0381; 20% 1.0810;
30% 1.1270; 40% 1.1764; 50% 1.2296; 60% 1.2865; 70% 1.3471; 76% 1.3854. nD20 of
10% soln 1.34783. Sucrose does
not reduce Fehling's soln, form an osazone, or show mutarotation.
It is hydrolyzed to glucose and fructose by dil acids and by invertase,
a yeast enzyme. Upon hydrolysis the optical rotation falls and is
negative when the hydrolysis is complete. The mixture of glucose
and fructose is known as “invert sugar.” Sucrose is fermentable, but
resists bacterial decompn when in high concentrations.Caution
Potential symptoms of overexposure
are irritation of eyes, skin, upper respiratory system; cough. See NIOSH Pocket Guide to Chemical
Hazards (DHHS/NIOSH 97-140, 1997) p 288.Use
Sweetening agent and food. Starting material in
the fermentative production of ethanol, butanol, glycerol, citric
and levulinic acids. Pharmaceutic aid (flavor, preservative, antioxidant
in the form of invert sugar, demulcent, substitute for glycerol, granulation
agent and excipient for tablets, coating for tablets). In the plastics
and cellulose industry, in rigid polyurethane foams, manuf of ink
and of transparent soaps. Optical rotation standard.
