8905. Sulfadimethoxine
Nomenclature
CAS number: 122-11-2
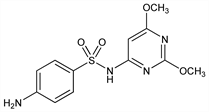
4-Amino-N-(2,6-dimethoxy-4-pyrimidinyl)benzenesulfonamide; N1-(2,6-dimethoxy-4-pyrimidinyl)sulfanilamide; 2,6-dimethoxy-4-sulfanilamidopyrimidine; 2,4-dimethoxy-6-sulfanilamido-1,3-diazine; 6-sulfanilamido-2,4-dimethoxypyrimidine; 2,6-dimethoxy-4-(p-aminobenzenesulfonamido)pyrimidine; Albon (Pfizer); Sulforal (Med-Pharmex); Ultrasulfon (Streuli). C12H14N4O4S; mol wt 310.33.
C 46.44%, H 4.55%, N 18.05%, O 20.62%, S 10.33%.
Description and references
Prepn: Bretschneider, Kltzer, US 2703800 (1955 to Oesterr. Stickstoffwerke); eidem, Monatsh. Chem. 87, 136 (1956); Langley, GB 866843 (1961 to I.C.I.); Bretschneider et al., Monatsh. Chem. 92, 128 (1961); US 3127398 (1964 to Hoffmann-La Roche); Shepherd et al., J. Org. Chem. 26, 2764 (1961). Toxicity data: Seki et al., Arzneim.-Forsch. 15, 1441 (1965). Toxicological, chemotherapeutic and pharmacokinetic studies: Bhni et al., Chemotherapy 14, 195-226 (1969); see also Aviado et al., ibid. 37.
Properties
Crystals from dil alc, mp 201-203°. Sol in dil HCl and in aq solns of sodium carbonate. Soly in water at 37° (mg/100 ml): 4.6 at pH 4.10; 29.5 at pH 6.7; 58.0 at pH 7.06; 5170 at pH 8.71. LD50 orally in mice: >10 g/kg (Seki).Derivative
Sodium salt.Nomenclature
CAS number: 1037-50-9
Di-Methox (AgriLabs). C12H13N4NaO4S; mol wt 332.31.
C 43.37%, H 3.94%, N 16.86%, Na 6.92%, O 19.26%, S 9.65%.