8906. Sulfadoxine
Nomenclature
Description and references
Dihydropteroate synthetase inhibitor. Prepn: BE 618639; H. Bretschneider et al., US 3132139 (1962, 1964 both to Hoffmann-La Roche). Toxicological, chemotherapeutic and pharmacokinetic studies: E. Bhni et al., Chemotherapy 14, 195-226 (1969). Comprehensive description: V. K. Kapoor, Anal. Profiles Drug Subs. 17, 571-605 (1988). Enzyme inhibition in vitro: Y. Zhang, S. R. Meshnick, Antimicrob. Agents Chemother. 35, 267 (1991). Determn in plasma using supercritical fluid chromatography: S. I. Bhoir et al., J. Chromatogr. B 757, 39 (2001). Clinical trial of combination with pyrimethamine, q.v., for malaria: C. V. Plowe et al., Br. Med. J. 328, 545 (2004).
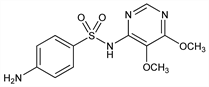
Properties
Crystals from 50% aq alc, mp 190-194°. Practically insol in ether. Very slightly sol in water, slightly sol in alcohol, methanol. Sol in dilute mineral acids, solutions of alkali hydroxides and carbonates. LD50 in mice (microcrystals, mg/kg): 5200 orally, 2900 s.c., 2900 i.p. (Bhni).Derivative
Mixture with trimethoprim.Nomenclature
Therapeutic Category
Antibacterial; antimalarial.
Therapeutic Category (Veterinary)
Antibacterial.Keywords
Antibacterial (Synthetic); Sulfonamides; Antimalarial