8908. Sulfaguanidine
Nomenclature
CAS number: 57-67-0
4-Amino-N-(aminoiminomethyl)benzenesulfonamide; 4-amino-N-(diaminomethylene)benzenesulfonamide; N1-amidinosulfanilamide; N1-guanylsulfanilamide; p-aminobenzenesulfonylguanidine; sulfanilylguanidine; RP-2275; Diacta (Nicholas); Ganidan (Specia); Guanicil (Cilag); Resulfon (Nordmark); Shigatox (Fawns & McAllan). C7H10N4O2S; mol wt 214.24.
C 39.24%, H 4.70%, N 26.15%, O 14.94%, S 14.97%.
Description and references
Prepd by condensing acetylsulfanilyl chloride with guanidine nitrate in the presence of much NaOH in water-acetone: Winnek, US 2218490 (1940); US 2229784 (1941); US 2233569 (1941); or by fusing N4-acetylsulfanilamide and dicyanodiamide: Haworth, Rose, GB 551524 (1943). Additional syntheses: ACS Monograph Series no. 106, entitled “Sulfonamides and Allied Compounds,” E. H. Northey, Ed. (Reinhold, New York, 1948). Revised structure: G. R. Sullivan, J. D. Roberts, J. Org. Chem. 42, 1095 (1977). Review of pharmacology: E. K. Marshall, Jr. et al., Bull. Johns Hopkins Hosp. 67, 163-188 (1940); E. Pick, J. Mt. Sinai Hosp. 10, 343-354 (1943).
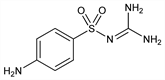
Derivative
Monohydrate.Properties
Needles. When anhydr, mp 190-193°. One gram dissolves in about 1000 ml water at 25° and in about 10 ml at 100°. Sparingly sol in alcohol or acetone. Freely sol in dil mineral acids. Insol in NaOH solns at room temp.Therapeutic Category
Antibacterial.
Therapeutic Category (Veterinary)
Antimicrobial. In enteric infections.Keywords
Antibacterial (Synthetic); Sulfonamides