Nomenclature
CAS number: 5329-14-6
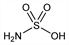
Amidosulfonic acid.
H
3NO
3S; mol wt 97.09.
H 3.11%, N 14.43%, O 49.44%, S 33.03%.
Description and references
Obtained from chlorosulfonic acid and ammonia,
or by heating urea with H2SO4. Purification:
Sisler et al., Inorg. Synth. 2, 178 (1946). Toxicity data: Ambrose, J. Ind. Hyg. Toxicol. 25, 26 (1943). Reviews: Audrieth et al., Chem. Rev. 26, 49 (1940); Burton, Nickless, “Amido-
and Imido-Sulfonic Acids” in Inorganic Sulphur Chemistry, G. Nickless, Ed. (Elsevier, New York, 1968) pp 607-627, 661-667;
E. B. Bell in Kirk-Othmer Encyclopedia of
Chemical Technology vol. 21 (Wiley-Interscience,
New York, 3rd ed., 1983) pp 949-960. Brief review of synthetic applications:
B. Wang, Synlett 2005, 1342-1343.
Properties
Orthorhombic crystals. d 2.15. mp ≈205° (dec). Corrosive. Stable when dry but in soln slowly hydrolyzes forming ammonium
bisulfate. Sol in 6.5 parts water at 0°, in about 2 parts water at
80°. Sulfuric acid decreases soly in water. Sparingly sol in alcohol,
methanol; slightly sol in acetone. Insol in ether. Freely sol in
nitrogenous bases, e.g., liquid ammonia, also in nitrogen contg organic
solvents, e.g., pyridine, formamide, dimethylformamide. Immiscible
with toluene, THF. A strong acid; pH of a 1% soln at 25° 1.18. Can
be titrated with bases by means of indicators showing color change
between pH 4.5 to 9. MLD orally in rats: 1.6 g/kg (Ambrose).Caution
Moderately irritating to skin, mucous
membranes.Use
As standard in alkalimetry and acidimetry; in acid
cleaning; in nitrite removal; in chlorine stabilization for use in
swimming pools, cooling towers, paper mills. Solid-acid catalyst
in organic synthesis. The acid or its ammonium salt has been recommended
for flameproofing fabrics and wood. Metal salts are used in electroplating.
Ammonium sulfamate,
q.v., is also widely used as a weed killer.
