8923. Sulfamidochrysoidine
Nomenclature
Description and references
Prototype compd leading to the development of sulfonamide antibacterials. Prepn: F. Mietzsch, J. Klarer, DE 607537 (1935 to I. G. Farbenind.); eidem, US 2085037 (1937 to Winthrop). Antibacterial activity in vivo: G. Domagk, Dtsch. Med. Wochenschr. 61, 573 (1935); (for English translation see Rev. Infect. Dis. 8, 163 (1986)). Identification of sulfanilamide as active metabolite: J. Tréfouel et al., C. R. Seances Soc. Biol. Ses Fil. 120, 756 (1935); L. Colebrook et al., Lancet 2, 1323 (1936). Polymorphic forms in commercial prepns: L. Kofler, A. Kofler, Monatsh. Chem. 81, 321 (1950). Use to visualize carbonic anhydrase during isoelectric focusing in polyacrylamide gels: W. Siffert et al., J. Biochem. Biophys. Methods 8, 331 (1983). Historical review: M. H. Bickel, Gesnerus 45, 67-86 (1988).
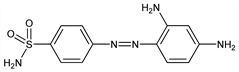
Derivative
Hydrochloride.Nomenclature
Properties
Orange-red crystals, mp 248-250°. One gram dissolves in 400 ml water; much more sol in hot water. Sol in alcohol, acetone, fats, oils.Use
Specific stain for carbonic anhydrase in polyacrylamide gels.Therapeutic Category
Antibacterial.
Keywords
Antibacterial (Synthetic); Sulfonamides