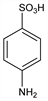
8926. Sulfanilic Acid
Nomenclature
CAS number: 121-57-3
4-Aminobenzenesulfonic acid; p-anilinesulfonic acid. C6H7NO3S; mol wt 173.19.
C 41.61%, H 4.07%, N 8.09%, O 27.71%, S 18.51%.
Description and references
Readily prepd from aniline and sulfuric acid: I. G. Farben, FIAT Final Rept. 1313 (I), 255 (1948); Fierz-David, Blangey, Fundamental Processes of Dye Chemistry (Interscience, New York, 1949) pp 126-128; A. I. Vogel, Practical Organic Chemistry (Longmans, London, 3rd ed., 1959) p 586; R. Q. Brewster et al., Unitized Experiments in Organic Chemistry (Van Nostrand, Princeton, 2nd ed., 1964) pp 162-163. Improved procedure: Jacobs et al., Ind. Eng. Chem. 35, 321-323 (1943).
Derivative
Monohydrate.Nomenclature
CAS number: 6101-32-2
Properties
Orthorhombic plates from water (very slow crystn can yield the dihydrate), becomes anhydr at around 100°. Dec without melting at ≈288° (also reported as >360°). pK2 (25°): 3.23. Slowly sol in water: ≈1% at 20°, 1.45% at 30°, 1.94% at 40° (w/w anhydr). Almost insol in ethanol, benzene, ether. Slightly sol in hot methanol.Derivative
Sodium salt dihydrate.Nomenclature
CAS number: 6106-22-5
C6H6NNaO3S.2H2O; mol wt 231.20.
C 31.17%, H 4.36%, N 6.06%, Na 9.94%, O 34.60%, S 13.87%.
Properties
Orthorhombic bipyramidal plates. Freely sol in water, sol in hot methanol.Derivative
Zinc salt tetrahydrate.Nomenclature
CAS number: 31884-76-1
Zinc sulfanilate tetrahydrate; Nizin (Broemmel). C12H12N2O6S2Zn.4H2O; mol wt 481.83.
C 29.91%, H 4.18%, N 5.81%, O 33.21%, S 13.31%, Zn 13.58%.
Derivative
Amide see Sulfanilamide.Use
Manuf various dyes and organic chemicals; as reagent in anal. chemistry (Ehrlich's reagent, detn of nitrites).Therapeutic Category
Antibacterial.
Keywords
Antibacterial (Synthetic); Sulfones