8925. Sulfanilamide
Nomenclature
Description and references
Active metabolite of the antibacterial dye, sulfamidochrysoidine, q.v. Prepn: P. Gelmo, J. Prakt. Chem. 77, 369 (1908); A. Galat, Ind. Eng. Chem. 36, 192 (1944); Hurdis, Yang, J. Chem. Educ. 46, 697 (1969). Large-scale process: F. Mietzsch, J. Klarer, US 2132178; eidem et al., US 2276664 (1938, 1942 both to Winthrop). Antibacterial activity: J. Tréfouel et al., C. R. Seances Soc. Biol. Ses Fil. 120, 756 (1935); and clinical efficacy: L. Colebrook, A. W. Purdie, Lancet 233, 1291 (1937). Toxicity study: E. K. Marshall, Jr. et al., J. Am. Med. Assoc. 110, 252 (1938). Historical review: M. H. Bickel, Gesnerus 45, 67-86 (1988).
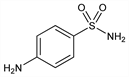
Properties
Crystals from boiling water, mp 164.5-166.5°. Neutral to litmus. pH (0.5% aq soln): 5.8-6.1. uv max: 255, 312 nm. One gram dissolves in about 37 ml alcohol; in about 5 ml acetone; in about 2 ml boiling water. Sol in glycerol, HCl, solns of K and Na hydroxides. Practically insol in chloroform, ether, benzene. LD50 orally in mice: 3.8 g/kg (Marshall).Derivative
N4-Acetylsulfanilamide.Nomenclature
Description and references
Major metabolite of sulfanilamide: E. K. Marshall, Jr. et al., Science 85, 202 (1937).
Properties
Needles from water, mp 219°.Therapeutic Category
Antibacterial.
Therapeutic Category (Veterinary)
Antibacterial.Keywords
Antibacterial (Synthetic); Sulfonamides