8937. Sulfapyrazine
Nomenclature
CAS number: 116-44-9
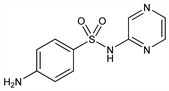
4-Amino-N-(pyrazinyl)benzenesulfonamide; N1-2-pyrazinylsulfanilamide; 2-sulfanilamidopyrazine. C10H10N4O2S; mol wt 250.28.
C 47.99%, H 4.03%, N 22.39%, O 12.79%, S 12.81%.
Description and references
Prepd by reacting acetylsulfanilyl chloride with 2-aminopyrazine in pyridine, followed by hydrolysis and neutralization: Ellingson, US 2420703 (1947). Biological properties: Ghione et al., Chemotherapia 6, 344 (1962).
Properties
Crystals, dec 250-254°. Very slightly sol in alc; slightly sol in acetone. Sol in aq solns of sodium, potassium, and barium hydroxides, in ammonia water, and in dil and concd mineral acid solns. Practically insol in water (5 mg/100 ml at 25°, and 5.2 mg/100 ml at 37°).Derivative
Sodium salt monohydrate.C10H9N4NaO2S.H2O; mol wt 290.27.
C 41.38%, H 3.82%, N 19.30%, Na 7.92%, O 16.54%, S 11.05%.
Properties
Bitter powder. Freely soluble in water (1 g/3.33 ml at 25°). Alkaline to litmus. pH of 10% aq soln 9.1. Very sol in acetone, slightly sol in alc. Insol in ether, chloroform. Aq solns of sulfapyrazine sodium may absorb carbon dioxide which causes precipitation of sulfapyrazine.Therapeutic Category
Antibacterial.
Therapeutic Category (Veterinary)
Antibacterial.Keywords
Antibacterial (Synthetic); Sulfonamides