8938. Sulfapyridine
Nomenclature
CAS number: 144-83-2
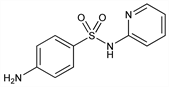
4-Amino-N-2-pyridinylbenzenesulfonamide; dagenan; N1-2-pyridylsulfanilamide; 2-sulfanilamidopyridine; 2-(p-aminobenzenesulfonamido)pyridine; M & B 693. C11H11N3O2S; mol wt 249.29.
C 53.00%, H 4.45%, N 16.86%, O 12.84%, S 12.86%.
Description and references
Prepn: A. J. Ewins, M. A. Phillips, GB 512145 (1939); eidem, US 2275354 (1942); R. Winterbottom, J. Am. Chem. Soc. 62, 160 (1940). Metabolism: R. Wien, J. Hampton, J. Pharmacol. Exp. Ther. 84, 211 (1945). Toxicology: R. Wien et al., ibid. 203. GC-MS determn in edible animal tissues: A. E. Mooser, H. Koch, J. AOAC Int. 76, 976 (1993). Efficacy and proposed mechanism of action in dermatological disease: O. J. Stone, Med. Hypotheses 31, 99 (1990). Clinical evaluation in ocular cicatricial pemphigoid: M. J. Elder et al., Br. J. Ophthalmol. 80, 549 (1996).
Properties
Crystals from alc, mp 190-191°. One gram dissolves in ≈3500 ml water, 440 ml alcohol, 65 ml acetone. Freely sol in dil mineral acids and in aq solns of KOH and NaOH. More sol in warm sugar solns than in water alone. The aq soln is neutral. LD50 orally in mice: 7.5 mg/g (Wien).Derivative
Sodium salt monohydrate.Nomenclature
CAS number: 127-57-1
Soluble sulfapyridine. C11H10N3NaO2S.H2O; mol wt 289.29.
C 45.67%, H 4.18%, N 14.53%, Na 7.95%, O 16.59%, S 11.08%.
Properties
On prolonged exposure to humid air it absorbs CO2, liberates sulfapyridine, becomes incompletely sol in water. One gram dissolves in ≈1.5 ml water, 10 ml alcohol. pH of 5% aq soln: 11.4. LD50 orally in mice: 2.7 mg/g (Wien).Therapeutic Category
In treatment of dermatitis herpetiformis; antibacterial.
Therapeutic Category (Veterinary)
Antibacterial.Keywords
Antibacterial (Synthetic); Sulfonamides; Dermatitis Herpetiformis Suppressant