8939. Sulfaquinoxaline
Nomenclature
CAS number: 59-40-5
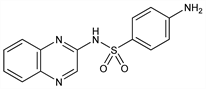
4-Amino-N-2-quinoxalinylbenzenesulfonamide; N1-(2-quinoxalinyl)sulfanilamide; 2-sulfanilamidoquinoxaline; N1-(2-quinoxalyl)sulfanilamide; sulfabenzpyrazine; Compd 3-120; S.Q. (Merck & Co.); Sulquin (Salsbury). C14H12N4O2S; mol wt 300.34.
C 55.99%, H 4.03%, N 18.65%, O 10.65%, S 10.68%.
Description and references
Prepd by treating 2-aminoquinoxaline with acetylsulfanilyl chloride in the presence of pyridine and hydrolyzing the resulting acetyl deriv: Weijlard et al., J. Am. Chem. Soc. 66, 1957 (1944); Weijlard, Tishler, US 2404199 (1946 to Merck & Co.). Anticoccidial spectrum: G. F. Mathis et al., Poult. Sci. 63, 1149 (1984). Evaluation of antibacterial and anticoccidial efficacy of mixture with trimethoprim: G. White, R. B. Williams, Vet. Rec. 113, 608 (1983); D. W. T. Piercy et al., ibid. 114, 60 (1984). Drug residue study in poultry muscle and liver: M. Patthy, J. Chromatogr. 275, 115 (1983). HPLC determn in rabbit plasma and urine: J. G. Eppel, J. J. Thiessen, J. Pharm. Sci. 73, 1635 (1984).
Properties
Minute crystals, mp 247-248°. uv max (pH 6.6 in H2O): 252, 360 nm (E1%1cm 1110, 275). Solubility in water at pH 7: 0.75 mg/100 ml; in 95% alcohol: 73 mg/100 ml; in acetone: 430 mg/100 ml. Sol in aq Na2CO3 and NaOH solns.Derivative
Sodium salt.Nomenclature
Aviochina (Agrimont-Vetem).C14H11N4NaO2S; mol wt 322.32.
C 52.17%, H 3.44%, N 17.38%, Na 7.13%, O 9.93%, S 9.95%.