8992. Sultamicillin
Nomenclature
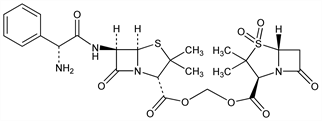
CAS number: 76497-13-7
(2S,5R,6R)-6-[[(2R)-Aminophenylacetyl]amino]-3,3-dimethyl-7-oxo-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxylic
acid [[[(2S,5R)-3,3-dimethyl-4,4-dioxido-7-oxo-4-thia-1-azabicyclo[3.2.0]hept-2-yl]carbonyl]oxy]methyl
ester; 1,1-dioxopenicillanoyloxymethyl 6-(d-α-amino-α-phenylacetamido)penicillanate; 6′-(2-amino-2-phenylacetamido)penicillanoyloxymethyl penicillanate
1,1-dioxide; CP-49952; VD-1827. C25H30N4O9S2; mol wt 594.66.
C 50.49%, H 5.08%, N 9.42%, O 24.21%, S 10.78%.
Description and references
Orally absorbed double ester of sulbactam and ampicillin, q.q.v. Prepn: E. C. Bigham, DE 3018590; eidem, US 4244951 (1980, 1981 both to Pfizer); and pharmacology: B. Baltzer et al., J. Antibiot. 33, 1183 (1980). HPLC determn and pharmacokinetics: H. J. Rogers et al., J. Antimicrob. Chemother. 11, 435 (1983). Clinical trial in pediatric otitis media: K. H. Chan et al., Pediatr. Infect. Dis. J. 12, 24 (1993). Review of antibacterial activity, pharmacokinetics and therapeutic efficacy: H. A. Friedel et al., Drugs 37, 491-522 (1989).
Properties
mp 190°.Derivative
Tosylate.Nomenclature
CAS number: 83105-70-8
Bacimex (Alter); Unacid PD oral (Pfizer); Unacim (Pfizer); Unasyn (Pfizer). C25H30N4O9S2.C7H8O3S; mol wt 766.86.
C 50.12%, H 4.99%, N 7.31%, O 25.04%, S 12.54%.
Therapeutic Category
Antibacterial.
Keywords
Antibacterial (Antibiotics); β-Lactams; Penicillins; Antibacterial Adjuncts; β-Lactamase Inhibitors