8997. Sumatriptan
Nomenclature
Description and references
Serotonin 5HT1-receptor agonist. Prepn: M. D. Dowle, I. H. Coates, DE 3320521; eidem, US 4816470; A. W. Oxford, GB 2162522 (1983, 1989, 1986 all to Glaxo). Receptor binding studies: P. P. A. Humphrey et al., Br. J. Pharmacol. 94, 1123 (1988); P. Schoeffter, D. Hoyer, Arch. Pharmacol. 340, 135 (1989). LC-MS determn in plasma: J. Oxford, M. S. Lant, J. Chromatogr. 496, 137 (1989). Clinical evaluations in migraine: A. Doenicke et al., Lancet 1, 1309 (1988); Subcutaneous Sumatriptan International Study Group, N. Engl. J. Med. 325, 316 (1991); in acute cluster headache: Sumatriptan Cluster Headache Study Group, ibid. 322. Review of pharmacology and clinical experience: S. J. Peroutka, Headache 30 (Suppl. 2), 554-560 (1990).
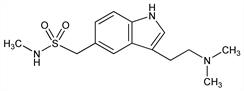
Properties
mp 169-171°.Derivative
Succinate.Nomenclature
Properties
mp 165-166°.Therapeutic Category
Antimigraine.
Keywords
Antimigraine; Serotonin Receptor Agonist