9020. Tabernanthine
Nomenclature
CAS number: 83-94-3
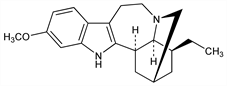
13-Methoxyibogamine. C20H26N2O; mol wt 310.43.
C 77.38%, H 8.44%, N 9.02%, O 5.15%.
Description and references
Indole alkaloid isolated from root of Tabernanthe iboga Baill., Apocynaceae: Delourme-Houdé, Ann. Pharm. Fr. 4, 30 (1946); Dickel et al., J. Am. Chem. Soc. 80, 123 (1958). Also in Tabernaemontana and Stemmadenia spp.; usually found in ibogaine mother liquors: Walls et al., Tetrahedron 2, 173 (1958). Isoln from genus Conopharingia, Apocynaceae: Renner, Prins, US 3008954 (1961 to Geigy). Structure: Bartlett et al., J. Am. Chem. Soc. 80, 126 (1958). Mass spectrum: Biemann, Friedmann-Spiteller, ibid. 83, 4805 (1961). Derivs: Taylor, US 2877229 (1959 to Ciba). Interaction with benzodiazepine receptors: J.-H. Trouvin et al., Eur. J. Pharmacol. 140, 303 (1987).
Properties
Needles or shiny leaflets from ethanol, mp 213.5-215°. Sublimes at 160° (0.005 mm pressure). [α]D20 40° (acetone). pKa 6.04 in 80% methylcellosolve. uv max (ethanol): 228, 271, 299 nm (log ε 4.53, 3.64, 3.77). Sol in alcohol, benzene, ether, chloroform. Practically insol in water.Derivative
Hydrochloride.C20H26N2O.HCl; mol wt 346.89.
C 69.25%, H 7.85%, N 8.08%, O 4.61%, Cl 10.22%.