Nomenclature
CAS number: 77-81-6
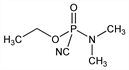
Dimethylphosphoramidocyanidic acid, ethyl ester; ethyl
N-dimethylphosphoramidocyanidate; dimethylamidoethoxyphosphoryl cyanide; GA.
C
5H
11N
2O
2P; mol wt 162.13.
C 37.04%, H 6.84%, N 17.28%, O 19.74%, P 19.10%.
Description and references
Nerve gas; potent cholinesterase inhibitor
similar in structure and activity to sarin and soman, q.q.v. Prepd
from dimethylamidophosphoryl dichloride and sodium cyanide in
the presence of ethanol: Holmstedt, Acta
Physiol. Scand. 25, Suppl. 90, 26 (1951).
Synthesis of dimethylamidophosphoryl dichloride: Michaelis, Ann. 326, 129 (1903). Alternate
synthetic route: B. C. Saunders, Some Aspects
of the Chemistry and Toxic Action of Organic Compounds Containing
Phosphorus and Fluorine (Cambridge, 1957) p 91. Toxicity
study: B. Holmstedt, Pharmacol. Rev. 11, 567 (1959). Brief review: Schrader, Die Entwicklung neuer insektizider Phosphorsure-Ester (Verlag Chemie, Weinheim, 1963) p 3; B. L. Harris in Kirk-Othmer Encyclopedia of Chemical Technology vol. 5 (Wiley-Interscience, New York, 4th ed., 1993)
pp 795-816.
Properties
Liquid. Fruity odor reminiscent of bitter almonds.
d 1.077. mp 50°. bp760 240°; bp10 120°; bp9 100-108°. nD20 1.4250. IR absorption: Acta
Chem. Scand. 5, 1179 (1951). Readily sol in
organic solvents. Miscible with water, but quickly hydrolyzed. Destroyed
by bleaching powder (chlorinated lime), but gives rise to cyanogen
chloride. Extremely poisonous. LD50 i.p. in mice:
0.6 mg/kg (Holmstedt).Caution
Potential symptoms of overexposure
by inhalation are constriction of pupils of the eye, difficulty breathing
followed by bronchial constriction, convulsions, death. Also toxic
due to absorption through skin and eyes. See Chem. Eng. News 31, 4676 (1953).Use
Chemical warfare agent.
