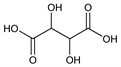
9069. dl-Tartaric Acid
Nomenclature
CAS number: 133-37-9
2,3-Dihydroxybutanedioic acid; racemic tartaric acid; racemic acid; dl-tartaric acid; resolvable tartaric acid; uvic acid; paratartaric acid; dl-Weinsure (German); Vogesensure (German); Traubensure (German). C4H6O6; mol wt 150.09.
C 32.01%, H 4.03%, O 63.96%.
Description and references
Probably never a natural product, although sometimes found in small amounts during wine-making. Prepn from l-tartaric acid by boiling with aq NaOH (meso-tartaric acid is obtained as a byproduct): Holleman, Org. Synth. 6, 82 (1926); coll. vol. I (2nd ed., 1941) p 497. Synthesis by oxidation of fumaric acid: Milas, Terry, J. Am. Chem. Soc. 47, 1412 (1925); Milas, Sussman, ibid. 58, 1302 (1936); US 2000213 (1935 to Standard Brands). From maleic acid: Church, Blumberg, Ind. Eng. Chem. 43, 1780 (1951).