Nomenclature
CAS number: 87-69-4
(2
R,3
R)-2,3-Dihydroxybutanedioic acid; ordinary tartaric acid; natural tartaric acid;
d-tartaric acid; (+)-tartaric acid; dextrotartaric acid;
l-2,3-dihydroxybutanedioic acid;
d-α,β-dihydroxysuccinic acid; Weinsure (German); Weinsteinsure (German).
C
4H
6O
6; mol wt 150.09.
C 32.01%, H 4.03%, O 63.96%.
Description and references
Dextrorotatory tartaric acid having a levo
configuration. Widely distributed in nature, classified as a fruit
acid. Occurs in many fruits, free and combined with potassium, calcium
or magnesium. Observed in antiquity as the acid potassium salt found
deposited as a fine crystalline crust during fermentation of grape
juice or tamarind juice and termed faecula (little yeast)
by the Romans. The derivation from Tartarus is of medieval,
alchemical origin. In modern processes the acid potassium tartrate
obtained during wine-making is first converted to calcium tartrate
which is then hydrolyzed to tartaric acid and calcium sulfate: Metzner, Chem. Eng. Prog. 43, 160 (1947);
several modifications, e.g., IT 490221 (1954 to Procedimenti Chimici), C.A. 50, 11607c
(1956). Extraction from tamarind pulp in about 10% yield: IN 52167 (1955), C.A. 50, 5249g (1956). Synthesis by hydroxylation of maleic
acid: Church, Blumberg, Ind. Eng. Chem. 43, 1780 (1951). Practically all of the l-tartaric
acid sold today is a byproduct of the wine industry. Monograph:
U. Roux, La Grande Industrie des Acides Organiques (Dounod, Paris, 1939). Example of a modern process: Dabul, US 3114770 (1963 to Orandi &
Massera).
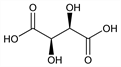
Properties
Monoclinic sphenoidal prisms, mp 168-170°. Stable to air and light.
Strong acid taste. Refreshing when in dil aq soln. d420 1.7598.
Odor of burnt sugar when heated to mp. [α]D20 +12.0° (c = 20
in H2O). Strong organic acid. At 25° pKa1 2.98; pKa2 4.34. pH of 0.1N soln:
2.2. Heat of combustion: 275.1 kcal/mol. Specific heat: 0.288
cal/g/°C at 21 to 51°; 0.296 at 0 to 99.6°. Dielectric constant 36.0
for 1200 cm waves. Freely sol in water. d415 of aq solns (w/w at 15°): 1% 1.0045; 10% 1.0469; 20% 1.0969; 30% 1.1505; 40% 1.2078; 50% 1.2696. Max soly in water in
g/100 ml at various temps: 0° = 115; 10° = 126; 20° = 139; 30° =
156; 40° = 176; 50° = 195; 60° = 217; 70° = 244; 80° = 273; 90° =
307; 100° = 343. One gram dissolves in 0.75 ml water at room temp,
in 0.5 ml boiling water, 1.7 ml methanol, 3 ml ethanol, 10.5 ml propanol,
250 ml ether. Also sol in glycerol. Insol in chloroform.Caution
Can cause local irritation. See Patty's Industrial Hygiene and
Toxicology vol. 2C, G. D. Clayton, F. E. Clayton,
Eds. (Wiley-Interscience, New York, 3rd ed., 1982) p 4937, 4943-4945.Use
In the soft drink industry, confectionery products,
bakery products, gelatin desserts, as an acidulant. In photography,
tanning, ceramics, manuf tartrates. The common commercial esters
are the diethyl and dibutyl derivs used for lacquers and in textile
printing. Pharmaceutic aid (buffering agent). Complexing agent.
