9077. Taxicins
Description and references
Acyl-free polyols from which the taxines are prepared by partial esterfication: Baxter et al., J. Chem. Soc. 1962, 2964. Structure of taxicin-I and -II: Eyre et al, Proc. Chem. Soc. London 1963, 271. Structure and stereochemical studies of taxicin-I and -II: Dukes et al., Tetrahedron Lett. 1965, 4765; J. Chem. Soc. C 1967, 448; Eyre et al., ibid. 452. Structure of O-cinnamoyltaxicin-II triacetate (taxinine): Kurono et al., Tetrahedron Lett. 1963, 2153; Nakanishi, Kurono, ibid. 1963, 2161; Ueda et al., ibid. 1963, 2167; Uyeo et al., J. Pharm. Soc. Jpn. 84, 762 (1964). Stereochemistry of taxinine: Kurono et al., Tetrahedron Lett. 1965, 1917; Shiro et al., Chem. Commun. 1966, 97.
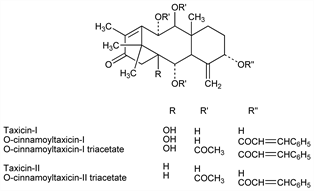
Derivative
Taxicin-I.Nomenclature
CAS number: 5308-89-4
[3S-(3α,4aα,5α,6α,11β,12α,12aβ)]-1,3,4,4a,5,6,7,11,12,12a-Decahydro-3,5,6,11,12-pentahydroxy-9,12a,13,13-tetramethyl-4-methylene-6,10-methanobenzocyclodecen-8(2H)-one. C20H30O6; mol wt 366.45.
C 65.55%, H 8.25%, O 26.20%.
Derivative
O-Cinnamoyltaxicin-I.Nomenclature
CAS number: 11034-45-0
C29H36O7; mol wt 496.59.
C 70.14%, H 7.31%, O 22.55%.
Properties
mp 233-234°. [α]D21 +285° (chloroform). uv max (alc): 282 nm (ε 28200).Derivative
O-Cinnamoyltaxicin-I triacetate.Nomenclature
CAS number: 13452-36-3
C35H42O10; mol wt 622.70.
C 67.51%, H 6.80%, O 25.69%.
Properties
Prisms from alc. mp 237-239°. [α]D18 +218° (chloroform).Derivative
Taxicin-II.Nomenclature
CAS number: 5308-90-7
C20H30O5; mol wt 350.45.
C 68.54%, H 8.63%, O 22.83%.
Derivative
O-Cinnamoyltaxicin-II triacetate.Nomenclature
CAS number: 3835-52-7
Taxinine. C35H42O9; mol wt 606.70.
C 69.29%, H 6.98%, O 23.73%.