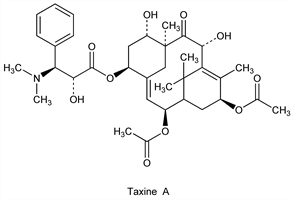
9078. Taxine
Nomenclature
Description and references
A mixture of alkaloids from needles and berries of the yew tree, Taxus baccata L., Taxaceae: Lucas, Arch. Pharm. 85, 145 (1856); Winterstein, Iatrides, Z. Physiol. Chem. 117, 240 (1921); Winterstein, Guyer, ibid. 128, 175 (1923); Callow et al., J. Chem. Soc. 1931, 2138. Structural studies of taxine-I: Baxter et al., ibid. 1962, 2964e; of taxine-II: Dukes et al., ibid. 1967, 448; Eyre et al., ibid. 452. Sepn of cryst taxine A and amorph taxine B from the amorph mixture: E. Graf, H. Bertholdt, Pharm. Zentralhalle 96, 385 (1957). Structure of taxine A: E. Graf et al., Ann. 1982, 376. Review: B. Lythgoe, The Alkaloids vol. X, R. H. F. Manske, Ed. (Academic Press, New York, 1968) pp 597-626. Toxicity data: Y. Tekol, Vet. Hum. Toxicol. 33, 337 (1991).