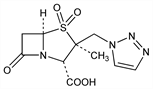
9083. Tazobactam
Nomenclature
Description and references
β-Lactamase inhibitor. Prepn: R. G. Micetich et al., EP 97446; eidem, US 4562073 (1984, 1985 both to Taiho); R. G. Micetich et al., J. Med. Chem. 30, 1469 (1987). Degradation in solution: T. Marunaka et al., Chem. Pharm. Bull. 36, 4478 (1988); in solid state: E. Matsushima et al., ibid. 4593. β-Lactamase inhibiting activity in comparison with clavulanic acid and sulbactam, q.q.v., vs aerobes: M. R. Jacobs et al., Antimicrob. Agents Chemother. 29, 980 (1986); vs anaerobes: P. C. Appelbaum et al., ibid. 30, 789. HPLC determn in biological materials: T. Marunaka et al., J. Chromatogr. 431, 87 (1988). Clinical trial in combination with piperacillin, q.v.: I. M. Gould et al., Drugs Exp. Clin. Res. 17, 187 (1991).
Derivative
Sodium salt.Nomenclature
Properties
Amorphous solid, mp >170° (dec).Derivative
Combination of sodium salt with piperacillin sodium.Nomenclature
Tazocilline (Wyeth); Tazocin (Wyeth); Zosyn (Wyeth).Therapeutic Category
In combination with β-lactam antibiotics as antibacterial.
Keywords
Antibacterial Adjuncts; β-Lactamase Inhibitors