9142. Temsirolimus
Nomenclature
CAS number: 162635-04-3
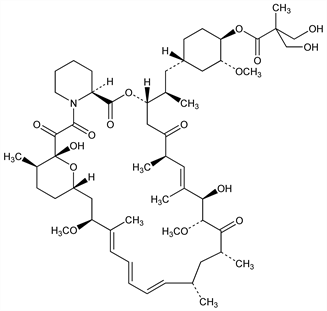
Rapamycin 42-[3-Hydroxy-2-(hydroxymethyl)-2-methylpropanoate]; (3S,6R,7E,9R,10R,12R,14S,15E,17E,19E,21S,23S,26R,27R,34aS)-9,10,12,13,14,21,22,23,24,25,26,27,32,33,34,34a-hexadecahydro-9,27-dihydroxy-3-[(1R)-2-[(1S,3R,4R)-hydroxy-3-methoxycyclohexyl]-1-methylethyl]-10,21-dimethoxy-6,8,12,14,20,26-hexameth-3H-pyrido[2,1c][1,4]oxaazacyclohentriacontine-1,5,11,28,29(4H,6H,31H)-pentone 4′-[2,2-bis(hydroxymethyl)propionate]; rapamycin 42-ester with 2,2-bis-(hydroxymethyl)propionic acid; CCI-779; Torisel (Wyeth). C56H87NO16; mol wt 1030.29.
C 65.28%, H 8.51%, N 1.36%, O 24.85%.
Description and references
Ester analog of rapamycin, q.v.; selectively inhibits mammalian target of rapamycin (mTOR). Prepn: J. S. Skotnicki et al., US 5362718 (1994 to Am. Home Prod.). Lipase-catalyzed synthesis from rapamycin: J. Gu et al., Org. Lett. 7, 3945 (2005). Clinical pharmacology: E. Raymond et al., J. Clin. Oncol. 22, 2336 (2004). Clinical study in advanced refractory renal cell carcinoma: M. B. Atkins et al., ibid. 909. Clinical evaluation in glioblastoma multiforme: E. Galanis et al., J. Clin. Oncol. 23, 5294 (2005); in breast cancer: S. Chan et al., ibid. 5314; in mantle cell lymphoma: T. E. Witzig et al., ibid. 5347.
Properties
White to off-white powder. Sol in alcohol. Practically insol in water. Soly is independent of pH. Lipophilic.Therapeutic Category
Antineoplastic.
Keywords
Antineoplastic; Antibiotics and Analogs