9146. Tenofovir
Nomenclature
Description and references
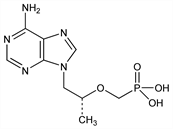
Acyclic phosphonate nucleotide analog; reverse transcriptase inhibitor. Prepn: I. Rosenberg et al., Collect. Czech. Chem. Commun. 53, 2753 (1988); A. Holy et al., ibid. 60, 1390 (1995); L. M. Schultze et al., Tetrahedron Lett. 39, 1853 (1998). Prepn of ester prodrug: M. N. Arimilli et al., Antivir. Chem. Chemother. 8, 557 (1997); idem et al., WO 9804569; eidem, US 5922695 (1998, 1999 both to Gilead Sci.). Antiretroviral activity in vitro: J. Balzarini et al., Biochem. Biophys. Res. Commun. 219, 337 (1996). Metabolism and pharmacokinetics of prodrugs: J.-P. Shaw et al., Pharm. Res. 14, 1824 (1997). Clinical trial in HIV-infected patients: J. E. Gallant et al., J. Am. Med. Assoc. 292, 191 (2004). Review of pharmacology and clinical efficacy: P. A. Pham, J. E. Gallant, Expert Opin. Drug Metab. Toxicol. 2, 459-469 (2006).
Properties
Crystals from boiling water + ethanol, mp 279°. [α]D +21° (c = 1 in 0.1M HCl).Derivative
Bis(isopropyloxycarbonyloxymethyl) ester.Nomenclature
Properties
Log P (1-octanol/aqueous phosphate buffer): 1.3 (pH 6.5).Derivative
Disoproxil fumarate.Nomenclature
Properties
White to off-white crystalline powder. Log P (octanol/phosphate buffer) at 25°: 1.25 (pH 6.5). Soly in distilled water (25°): 13.4 mg/ml.Therapeutic Category
Antiretroviral.
Keywords
Reverse Transcriptase Inhibitor; Antiretroviral; Reverse Transcriptase Inhibitors; Nucleosides/Nucleotides