9171. α-Terpineol
Nomenclature
CAS number: 98-55-5
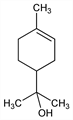
α,α,4-Trimethyl-3-cyclohexene-1-methanol; p-menth-1-en-8-ol. C10H18O; mol wt 154.25.
C 77.87%, H 11.76%, O 10.37%.
Description and references
Terpineol exists as three isomers, α-, β-, and γ-terpineol: J. L. Simonsen, The Terpenes vol. I (University Press, Cambridge, 2nd ed., 1947) pp 256-274. Isoln of d-α-terpineol from petitgrain oil: Walbaum, Hüthig, J. Prakt. Chem. 67, 322 (1903). Isoln from l-α-terpineol from long leaf pine oil: Teeple, J. Am. Chem. Soc. 30, 412 (1908). Isoln of dl-α-terpineol from cajeput oil: Voiry, Compt. Rend. 106, 1540 (1888). Synthesis of d-α-terpineol: Cologne, Crabalona, Bull. Soc. Chim. Fr. 1960, 102. Synthesis of l-α-terpineol: eidem, ibid. 1959, 1505. Stereochemistry: Henbest, McElkinney, J. Chem. Soc. 1959, 1834. Review: Wagner, Manuf. Chem. 22, 98, 153 (1951).
Derivative
d-Form.Properties
Liquid. bp4.5 81-82°; bp731 206-207°. d420 0.9338. nD20 1.4818. [α]D20 +92.45°. Solidifies at 31°.Derivative
Phenylurethan.C17H23NO2; mol wt 273.37.
C 74.69%, H 8.48%, N 5.12%, O 11.71%.
Properties
Crystals from petr ether, mp 111°. [α]D20 +30.50° (benzene).Derivative
l-Form.Properties
Liquid. bp5 80-81.5°. d420 0.935. nD20 1.4820. [α]D20 -100° (c = 20 in alc). Solidifies at 36.4°.Derivative
Dinitrobenzoate.C17H20N2O6; mol wt 348.35.
C 58.61%, H 5.79%, N 8.04%, O 27.56%.
Properties
Crystals from alcohol, mp 101.5°. [α]D20 -31° (c = 9.5 in carbon tetrachloride).Derivative
dl-Form.Properties
Liquid. bp3 85°; bp752 218.8-219.4°, d15 0.9386. nD20 1.4831.Derivative
Phenylurethan.C17H23NO2; mol wt 273.37.
C 74.69%, H 8.48%, N 5.12%, O 11.71%.
Properties
Crystals, mp 113°.Use
Perfumes; denaturing fats for soap manufacture.Therapeutic Category
Antiseptic.
Keywords
Antiseptic/Disinfectant