9173. Tertatolol
Nomenclature
Description and references
Nonselective β-adrenergic blocker. Prepn: C. Malen, M. Laubie, DE 2115201; eidem, US 3960891 (1971, 1976 both to Sci. Union et Cie). Pharmacology in animals: M. Laubie et al., Arch. Int. Pharmacodyn. Ther. 201, 323, 334 (1973); B. R. Walker et al., J. Cardiovasc. Pharmacol. 7, 1193 (1985). Synergistic effect with indapamide, q.v.: E. Marmo et al., Drugs Exp. Clin. Res. 11, 709 (1985). GC-MS determn in plasma and urine: S. Staveris et al., J. Chromatogr. 339, 97 (1985). Pharmacology in humans and mechanism of action study: A. De Blasi et al., Clin. Pharmacol. Ther. 39, 245 (1986). Preliminary evaluation in hypertension: J. P. Degaute et al., Am. J. Hypertens. 1, 263S (1988).
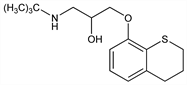
Properties
Crystals from hexane, mp 70-72°.Derivative
Hydrochloride.Nomenclature
Properties
Crystals from acetonitrile, mp 180-183°. pKa 9.8. LD50 in rats, mice (mg/kg): 40, 37 i.v.; 90, 120 i.p. (Laubie).Therapeutic Category
Antihypertensive.
Keywords
β-Adrenergic Blocker; Antihypertensive; Aryloxypropanolamine Derivatives