9180. Testosterone
Nomenclature
Description and references
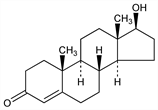
Principal hormone of the testes, produced by the interstitial cells. Major circulating androgen; converted by 5α-reductase in androgen-dependent target tissues to 5α-dihydrotestosterone which is required for normal male sexual differentiation. Also converted by aromatization to estradiol, q.v. Isoln from bull testes: David et al., Z. Physiol. Chem. 233, 281 (1935). Prepn from cholesterol and confirmation of structure: A. Butenandt, G. Hanisch, Ber. 68, 1859 (1935); eidem, Z. Physiol. Chem. 237, 89 (1935); from dehydroandrosterone: L. Ruzicka, A. Wettstein, Helv. Chim. Acta 18, 1264 (1935); from mixed esters: L. Ruzicka et al., ibid. 1478. Crystal structure: P. J. Roberts et al., J. Chem. Soc. Perkin Trans. 2 1973, 1978. Historical review: J. M. Hoberman, C. E. Yesalis, Sci. Am. 272, 76-81 (Feb. 1995). Review of role in aging males: F. E. Kaiser, J. E. Morley, Neurobiol. Aging 15, 559-563 (1994); of clinical relevance in females: R. S. Rittmaster, Am. J. Med. 98, Suppl. 1A, 17S-21S (1995).
Properties
Needles from dil acetone, mp 155°. [α]D24 +109° (c = 4 in alc). uv max: 238 nm. Insol in water. Sol in alcohol, ether, and other organic solvents.Derivative
Acetate.Nomenclature
Properties
mp 140-141°.Derivative
17β-Cyclopentanepropionate.Nomenclature
Description and references
Pharmacology: A. C. Ott et al., J. Clin. Endocrinol. Metab. 12, 15 (1952).
Properties
Crystals, mp 101-102°. [α]D25 +87° (CHCl3). Sol in oils.Derivative
Enanthate.Nomenclature
Description and references
Prepn: Junkmann et al., US 2840508 (1958 to Schering AG). Comprehensive description: K. Florey, Anal. Profiles Drug Subs. 4, 452-465 (1975).
Properties
Crystals, mp 36-37.5°.Derivative
Propionate.Nomenclature
Description and references
Prepn: K. Miescher et al., US 2109400 (1938 to Soc. Chem. Ind. Basel).
Properties
Stout prisms from alcohol + water, mp 118-122°. [α]D25 +83 to +90° (100 mg in 10 ml dioxane). Insol in water. Freely sol in alcohol, ether, pyridine, in other organic solvents. Sol in vegetable oils.Derivative
Undecanoate.Nomenclature
Note
This is a controlled substance (anabolic steroid): 21 CFR, 1308.13, as defined in 1300.01.Therapeutic Category
Androgen.
Therapeutic Category (Veterinary)
Androgen.Keywords
Androgen