9213. Tetrahydrofurfuryl Alcohol
Nomenclature
CAS number: 97-99-4
Tetrahydro-2-furanmethanol; tetrahydro-2-furancarbinol; tetrahydro-2-furylmethanol; THFA. C5H10O2; mol wt 102.13.
C 58.80%, H 9.87%, O 31.33%.
Description and references
Prepn by catalytic hydrogenation of furfuryl alcohol: Lukes, Nelson, J. Org. Chem. 21, 1096 (1956). Manuf by catalytic hydrogenation of furfural or furfuryl alcohol: Dunlop, Schegulla, US 2838523 (1958 to Quaker Oats). Occurs in two isomeric forms: d-isomer (levorotatory), l-isomer (dextrorotatory). Abs config: Gagnaire, Butt, Bull. Soc. Chim. Fr. 1961, 312; Hartman, Barker, J. Org. Chem. 29, 873 (1964). Review: A. P. Dunlop, F. N. Peters, The Furans (Reinhold, New York, 1953).
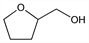
Properties
Liquid. Hygroscopic. d2020 1.0543; d2424 1.0511; d3131 1.0450. Melts below -80°. bp760 178°. nD20 1.4520; nD25 1.4499. Flash pt, open cup: 183°F (84°C). Flammability in air: Upper limit 9.7% by vol, lower limit 1.5% by vol. Heat capacity at 30-37°: 0.432 cal/g/°C. Heat of combustion at constant vol: 708.6 cal/g mole. Viscosity at 20°: 6.24 cP. Surface tension at 25°: 37 dyn/cm. Octane no. 82.5. Evaporation rate: 7 (n-butyl acetate = 100). Kauributanol value 71.5. Dilution ratio (lacquer ingredients): 4.5. Dielectric constant at 23°: 13.6. Miscible with water, alcohol, ether, acetone, chloroform, benzene.Derivative
l-Isomer.Description and references
Prepn see Hartman, Barker, loc. cit.