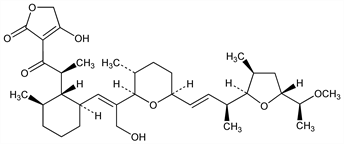
9248. Tetronasin
Nomenclature
Description and references
Polyether antibiotic produced by Streptomyces longisporoflavus NCIB 11426. Possesses a biosynthetically rare acid grouping in the form of an acyl tetronic acid moiety. Isoln and use in ruminants: D. H. Davies, G. L. F. Norris, GB 2027013; eidem, US 4279894 (1980, 1981 both to ICI). Physical data and crystal structure: D. H. Davies et al., Chem. Commun. 1981, 1073. Solution conformation and cation-binding properties: J. Grandjean, P. Laszlo, Tetrahedron Lett. 24, 3319 (1983). Synthetic studies: A. M. Doherty, S. V. Ley, ibid. 27, 105 (1986). Biosynthetic studies: J. M. Bulsing et al., Chem. Commun. 1984, 1301; D. M. Doddrell et al., ibid. 1302; A. K. Demetriadou et al., ibid. 1985, 408. Antimicrobial activity: C. J. Newbold et al., Appl. Environ. Microbiol. 54, 544 (1988). Effect on gain efficiency in cattle: S. J. Bartle et al., J. Anim. Sci. 66, 1502 (1988).