9277. Thebainone
Nomenclature
CAS number: 467-98-1
7,8-Didehydro-4-hydroxy-3-methoxy-17-methylmorphinan-6-one; thebainone-A. C18H21NO3; mol wt 299.36.
C 72.22%, H 7.07%, N 4.68%, O 16.03%.
Description and references
Prepn from thebaine, codeinone or β-ethylthiocodide: Morris, Small, J. Am. Chem. Soc. 56, 2159 (1934). Earlier references and discussion of structure: Small, Lutz, “Chemistry of the Opium Alkaloids” in U.S. Public Health Reports Suppl. No. 103 (Washington, 1932). About anomalies in nomenclature and difference from metathebainone see Henry, Plant Alkaloids (London, 1939) p 249. Description of all thebainones: K. W. Bentley, The Chemistry of the Morphine Alkaloids (Oxford, 1954) p 219.
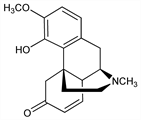
Properties
Crystals from ethyl acetate, mp 146°. [α]D28 -47° (c = 1.16 in 95% alc). One gram dissolves in 250 ml water, about 120 ml boiling water. Sol in chloroform, benzene, acetone; sparingly sol in ether, alcohol, methanol.Derivative
Sesquihydrate.Properties
Crystals from water, mp 90°.Derivative
Methanolate.Properties
Crystals from methanol, mp 118°.Derivative
Hydrochloride.C18H21NO3.HCl; mol wt 335.83.
C 64.38%, H 6.60%, N 4.17%, O 14.29%, Cl 10.56%.
Properties
Crystals from alcohol, mp 256° (turns red at mp). [α]D30 -25° (c = 1.63).Derivative
Hydriodide.C18H21NO3.HI; mol wt 427.28.
C 50.60%, H 5.19%, N 3.28%, O 11.23%, I 29.70%.