9287. Thermorubin
Nomenclature
CAS number: 11006-83-0
Description and references
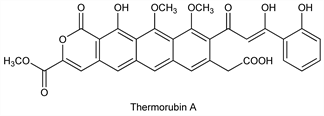
Antibiotic substances isolated from Thermoactinomyces antibioticus: Craveri et al., Clin. Med. 71, 511 (1964); eidem, DE 1180891; eidem, US 3300379 (1964, 1967 both to Lepetit). Structure of thermorubin A, the major component: Moppett et al., J. Am. Chem. Soc. 94, 3269 (1972); revised structure: F. Johnson et al., ibid. 102, 5580 (1980). Inhibition of bacterial protein synthesis: G. Pirali et al., Biochim. Biophys. Acta 366, 310 (1974). Studies on mechanism of action: F. Lin, A. Wishnia, Biochemistry 21, 477, 484 (1982).
Properties
Orange-red rosettes and needles from chloroform, darkens 190°, chars 300° without melting. [α]D25 -14° (c = 0.4 in dioxane). Absorption max [dioxane-cyclohexane (1:1)]: 300, 328, 414, 430 nm (E1%1cm 1041, 1066, 313, 332). Sol in dioxane, pyridine, tetrahydrofuran, DMF, DMSO, concd alkalies, concd H2SO4, glacial acetic acid; slightly sol in methanol, ethanol, butanol, ethyl acetate, acetone, chloroform, benzene, cyclohexane. Practically insol in water, ether, petr ether, hexane. LD50 i.p. in mice: 300 mg/kg (Craveri, 1967).Derivative
Diacetate.Properties
Crystals from ethyl acetate. Absorption max [0.1N HCl-dioxane (1:1)]: 337, 436, 462 nm (E1%1cm 1500, 113, 162).Derivative
Thermorubin A.C32H24O12; mol wt 600.53.
C 64.00%, H 4.03%, O 31.97%.