9295. Thiamine
Nomenclature
Description and references
Essential nutrient required for carbohydrate metabolism; also involved in nerve function. Biosynthesized by microorganisms and plants. Dietary sources include whole grains, meat products, vegetables, milk, legumes and fruit. Also present in rice husks and yeast. Converted in vivo to thiamine diphosphate, a coenzyme in the decarboxylation of α-keto acids. Chronic deficiency may lead to neurological impairment, beriberi, Wernicke-Korsakoff syndrome. Isoln from rice bran: B. C. P. Jansen, W. F. Donath, Chem. Weekbl. 23, 201 (1926). Structure: R. R. Williams, J. Am. Chem. Soc. 58, 1063 (1936); R. R. Williams, J. K. Cline, ibid. 1504; R. R. Williams et al., ibid. 59, 526 (1937). Review of syntheses: Knobloch in H. Vogel, Chemie und Technik der Vitamine vol. II (Stuttgart, 1953) pp 1-128. Toxicity data: D. Winter et al., Int. Z. Vitaminforsch. 37, 82 (1967). HPLC determn in foods, pharmaceuticals, body tissues: T. Kawaski, Methods Enzymol. 122, 15 (1986); in plasma and pharmacokinetics: H. Mascher, C. Kikuta, J. Pharm. Sci. 82, 56 (1993). Review of bioavailability, absorption, and role in nutrition: F. L. Iber et al., Am. J. Clin. Nutr. 36, 1067-1082 (1982). Reviews: “Thiamin: Twenty Years of Progress”, Ann. N.Y. Acad. Sci. 378, H. Z. Sable, C. J. Grubier, Eds. (1982) 470 pp; “Thiamin, Vitamin B1, Aneurin” in Vitamins, W. Friedrich, Ed. (de Gruyter, Berlin, 1988) pp 339-401.
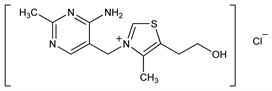
Derivative
Hydrochloride.Nomenclature
Description and references
Comprehensive description: K. A. M. Al-Rashood et al., Anal. Profiles Drug Subs. 18, 413-458 (1989).
Properties
Monoclinic plates in rosette-like clusters. Slight thiazole odor. Bitter taste. dec 248°. One gram dissolves in ≈1 ml water, 18 ml glycerol, 100 ml 95% alcohol, 315 ml abs alcohol; more sol in methanol. Sol in propylene glycol. Practically insol in ether, benzene, hexane, chloroform. pH of a 1% w/v soln in water 3.13; pH of a 0.1% w/v soln in water 3.58. On exposure to air of average humidity, the vitamin absorbs an amount of water corresponding to nearly one mol, forming a hydrate. LD50 in mice (mg/kg): 89.2 i.v.; 8224 orally (Winter).Derivative
Mononitrate.Nomenclature
Description and references
Prepn: R. J. Turner, G. J. Schmitt, US 2844579 (1958 to Am. Cyanamid).
Properties
Crystals, mp 196-200° (dec). Practically nonhygroscopic. pKa 4.8. Soly in water (g/100 ml): 2.7 (25°); ≈30 (100°). pH of 2% aq soln 6.5 to 7.1. More stable than the hydrochloride; suitable for enrichment of flours and feeds, multivitamin prepns.Therapeutic Category
Vitamin (enzyme cofactor).
Therapeutic Category (Veterinary)
Vitamin (enzyme cofactor).Keywords
Enzyme Cofactor; Vitamin/Vitamin Source; Vitamin B1