9296. Thiamine Diphosphate
Nomenclature
Description and references
Coenzyme required for the decarboxylation of α-keto acids. Most abundant form of thiamine in animal tissues. Identification as cofactor and enzymatic synthesis: K. Lohmann, P. Schuster, Biochem. Z. 294, 183 (1937). Chemical synthesis: Weijlard, Tauber, J. Am. Chem. Soc. 60, 2263 (1938); Weil-Malherbe, Biochem. J. 34, 980 (1940); Weijlard, J. Am. Chem. Soc. 63, 1160 (1941); Karrer, Viscontini, Helv. Chim. Acta 29, 711 (1946). LC determn in blood: J. W. I. Brunnekreeft et al., J. Chromatogr. 491, 89 (1989). Role in pathogenesis of Wernicke-Korsakoff Syndrome: R. F. Butterworth et al., Alcohol. Clin. Exp. Res. 17, 1084 (1993). Review of enzyme activity: J. Ullrich et al., Vitam. Horm. 28, 365 (1970); of mechanism of action: R. Kluger, Chem. Rev. 87, 863-876 (1987); M. Louloudi, N. Hadjiliadis, Coord. Chem. Rev. 135, 429-468 (1994).
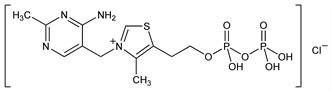
Derivative
Monohydrate.Properties
Crystals from alc contg some HCl, dec 240-244°. mp 238-240° from abs ethanol. uv max: 242 nm. Soluble in water. pH of 0.3% soln 2.23. The dry substance is very stable.Derivative
Free ester tetrahydrate.Description and references
Prepn: Wenz et al., US 2991284 (1961 to E. Merck).
Properties
Dec 220-225°.Derivative
Thiamine monophosphate.Nomenclature
Description and references
Prepd by sulfuric acid hydrolysis of thiamine diphosphate: Lohmann, Schuster, loc. cit.
Properties
Monohydrate; prisms from dil acetone or water, mp ≈200°. Freely sol in water.Therapeutic Category
Vitamin (enzyme cofactor).
Keywords
Enzyme Cofactor; Vitamin/Vitamin Source; Vitamin B1