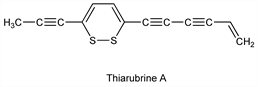
9304. Thiarubrines
Description and references
Class of dark red bioactive dithiacyclohexadiene polyacetylenes isolated from various plant sources. Isoln from Eriophyllum caespitosum Dougl., Compositae: F. Bohlmann, K.-M. Kleine, Ber. 98, 3081 (1965); and separation by HPLC: R. A. Norton et al., Phytochemistry 24, 356 (1985); from Ambrosia chamissonis (Less.) Greene: S. Ellis et al., ibid. 33, 224 (1993). Isoln of A from leaves of African medicinal plants of the genus Aspilia: E. Rodriguez et al., Experientia 41, 419 (1985). NMR spectra and biosynthetic study of A: M. L. Gomez-Barrios et al., Phytochemistry 31, 2703 (1992). Synthesis of A: M. Koreeda, W. Yang, J. Am. Chem. Soc. 116, 10793 (1994); of B: E. Block et al., ibid. 9403; of C: Y. Wang et al., J. Org. Chem. 63, 8644 (1998) . Antibiotic properties of A: G. H. N. Towers et al., Planta Med. 51, 225 (1985); photo-enhancement of activity by conversion to thiophenes: C. P. Constabel, G. H. N Towers, ibid. 55, 35 (1989). Antiviral properties of A: J. B. Hudson et al., ibid. 52, 51 (1986); photo-enhancement of activity against HIV: idem et al., Photochem. Photobiol. 57, 675 (1993).