Nomenclature
CAS number: 68-11-1
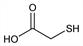
2-Mercaptoacetic acid; thioglycollic acid.
C
2H
4O
2S; mol wt 92.12.
C 26.08%, H 4.38%, O 34.74%, S 34.81%.
Description and references
Prepd by the action of sodium sulfhydrate on
sodium chloroacetate; by electrolysis of dithioglycollic acid (from
sodium sulfide and sodium chloroacetate). Toxicity study: Deichmann, Mergard, J. Ind.
Hyg. Toxicol. 30, 373 (1948).
Properties
Liquid, strong, unpleasant odor. Corrosive. Readily oxidized by air. d 1.325. mp 16.5°. bp29 123°. bp15 108°. bp5 96°. K1 at 25° = 2.1 × 104; K2 = 2.1 × 1011. Miscible with water, alcohol, ether, chloroform, benzene, and
many other organic solvents. LD50 orally in rats: 0.15 ml/kg (Deichmann, Mergard).Caution
Potential symptoms of overexposure
are irritation of eyes, skin, nose, throat; lacrimation, corneal damage;
skin burns, blisters. See NIOSH
Pocket Guide to Chemical Hazards (DHHS/NIOSH 97-140, 1997)
p 306.Use
Sensitive reagent for iron, molybdenum, silver,
tin. With ferric iron a blue color appears, and when an alkali hydroxide
is added to a soln contg ferrous salts and thioglycolic acid, a yellow
precipitate forms. Used in the manuf of thioglycolates. The ammonium
and sodium salts are commonly used for cold waving and the calcium
salt is a depilatory. The sodium salt also is used in bacteriology
in the prepn of thioglycolate media.
