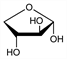
9381. d-Threose
Nomenclature
Description and references
From calcium d-xylonate by oxidation with H2O2: Ruff, Ber. 34, 1370 (1901). Improved procedure using strontium d-xylonate and ferric acetate catalyst: Hockett, J. Am. Chem. Soc. 57, 2260, 2265 (1935). From tetraacetyl-d-xylononitrile: Maquenne, Compt. Rend. 130, 1403 (1900); Ann. Chim. [7] 24, 404 (1901); Bonner, Roth, J. Am. Chem. Soc. 81, 5454 (1959); from monobenzylidene-d-arabitol: Steiger, Reichstein, Helv. Chim. Acta 19, 1016 (1939); from 1,1-diethylsulfonyl-d-threo-3,4,5-trihydroxypent-1-ene: Hough, Taylor, J. Chem. Soc. 1955, 1212; from d-galactose: Perlin, Brice, Can. J. Chem. 34, 541 (1956). Synthesis of dl-threose: Lake, Glattfeld, J. Am. Chem. Soc. 66, 1091 (1944); Schmid, Grob, Helv. Chim. Acta 32, 77 (1949); Sonogashira, Nakagawa, Bull. Chem. Soc. Jpn. 45, 2616 (1972).