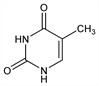
9398. Thymine
Nomenclature
CAS number: 65-71-4
5-Methyl-2,4(1H,3H)-pyrimidinedione; 5-methyluracil; 2,4-dihydroxy-5-methylpyrimidine. C5H6N2O2; mol wt 126.11.
C 47.62%, H 4.80%, N 22.21%, O 25.37%.
Description and references
A pyrimidine derivative; constituent of nucleic acids. Originally isolated from thymus nucleic acid: Levene, Z. Physiol. Chem. 39, 4 (1903). Prepn by heating 2-ethylmercapto-4-hydroxy-5-methylpyrimidine: Wheeler, Merriam, Am. Chem. J. 29, 478 (1903); 43, 29 (1910). From methylcyanacetylurea by catalytic reduction: Bergmann, Johnson, J. Am. Chem. Soc. 55, 1733 (1933). From β-methylmalic acid: Scherp, J. Am. Chem. Soc. 68, 912 (1946). Crystal structure of monohydrate: Gerdil, Acta Crystallogr. 14, 333 (1961). Review: Ts'o, “Bases, Nucleosides and Nucleotides” in Basic Principles in Nucleic Acid Chemistry vol. 1, P. O. P. Ts'o, Ed. (Academic Press, New York, 1974) pp 453-584.