9399. Thymol
Nomenclature
Description and references
Isolated by Neumann in 1719. Obtained from the essential oil of Thymus vulgaris L. and Monarda punctata L., Labiatae: Arppe, Ann. 58, 41 (1846); Meyer, Pharm. Ztg. 81, 192, 205 (1936). Also occurs in other volatile oils. Produced synthetically from p-cymene, piperitone, or m-cresol: Austerweil, GB 221227 (1923); Jennen, Verdroncken, Compt. Rend. 245, 183 (1957); Bottoms, US 2840616 (1958 to Natl. Cylinder Gas). Bactericidal activity: J. M. Schaffer, F. W. Tilley, J. Bacteriol. 14, 259 (1927). Mold elimination on surfaces: O. W. Richards, K. J. Hawley, J. Chem. Educ. 16, 6 (1939). In vitro antifungal activity: H. B. Myers, J. Am. Med. Assoc. 89, 1834 (1927). Effectiveness as antifungal preservative: M. Dersarkissian, M. Goodberry: Stud. Conserv. 25, 28 (1980). Use as clinical preservative: T. Z. Liu, Clin. Chem. 25, 336 (1979); T. Z. Liu et al., ibid. 27, 1144 (1981). Toxicity: P. M. Jenner et al., Food Cosmet. Toxicol. 2, 327 (1964).
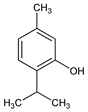
Properties
Crystals, mp 51.5°. bp ≈233°. Appreciably volatile at 100°; volatilizes in water vapors. Characteristic odor; pungent, somewhat caustic taste. d425 0.9699. nD20 1.5227; nD25 1.5204. One gram dissolves in ≈1000 ml water, 1 ml alcohol, 0.7 ml chloroform, 1.5 ml ether, 1.7 ml olive oil at 25°. Sol in glacial acetic acid, oils, fixed alkali hydroxides. LD50 orally in rats: 980 mg/kg (Jenner). Incompat: Acetanilide, antipyrine, camphor, monobromated camphor, chloral hydrate, menthol, quinine sulfate, salol, urethane, spirit nitrous ether; in triturations because of liquefaction.Derivative
Acetate.Nomenclature
Properties
Yellowish, oily liq; thymol odor. d0 1.009. bp 243.5-245.5°. Practically insol in water. Miscible with alcohol, benzene chloroform, ether.Caution
Mild irritant.Derivative
Carbonate.Nomenclature
Properties
White crystals; thymol odor; volatilizes with steam. mp 49°. Insol in water, acids, alkalies. Sol in hot alcohol, chloroform, ether, carbon tetrachloride.Use
For destroying mold; preserving documents, art objects and urine. Stabilizer (antioxidant) for trichloroethylene, halothane.Therapeutic Category
Antiseptic (topical); anthelmintic (Nematodes).
Therapeutic Category (Veterinary)
Has been used as anthelmintic, and as an antiseptic, external and internal.Keywords
Anthelmintic (Nematodes)