9428. Ticarcillin
Nomenclature
Description and references
Broad spectrum semi-synthetic antibiotic related to penicillin. Prepn: BE 646991; E. G. Brain, J. H. Nayler, US 3282926 (1964, 1966 to Beecham Group Ltd.). In vitro studies: H. C. Neu, E. B. Winshell, Antimicrob. Agents Chemother. 1970, 385; R. Sutherland et al., ibid. 390; N. J. Legakis, J. Papavassiliou, J. Antibiot. 28, 912 (1975). In vivo studies: P. Acred et al., Antimicrob. Agents Chemother. 1970, 396. Absorption and excretion: R. Sutherland, P. J. Wise, ibid. 402. Clinical pharmacology: V. Rodriguez et al., Antimicrob. Agents Chemother. 4, 31 (1973); R. D. Libke et al., Clin. Pharmacol. Ther. 17, 441 (1975). Review of pharmacology and therapeutic efficacy: R. N. Brogden et al., Drugs 20, 325-352 (1980).
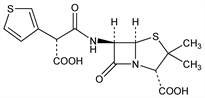
Derivative
Disodium salt.Nomenclature
Properties
Creamy-white hygroscopic non-crystalline powder. Readily sol in water (>100 g/100 ml water) giving a clear soln with pH between 6.0 and 8.0. Aq solns are relatively stable; acid solns relatively unstable (Sutherland).Therapeutic Category
Antibacterial.
Therapeutic Category (Veterinary)
Antibacterial.Keywords
Antibacterial (Antibiotics); β-Lactams; Penicillins