9429. Ticlopidine
Nomenclature
Description and references
Platelet aggregation inhibitor. Prepn: DE 2404308; A. R. J. Castaigne, US 4051141 (1974, 1977 both to Cent. Etudes Ind. Pharm.); E. Braye, US 4127580 (1978 to Parcor). Metabolism: P. Godard et al., Eur. J. Drug Metab. Pharmacokinet. 3, 67 (1978); eidem, ibid. 4, 133 (1979); A. Tuong et al., ibid. 6, 91 (1981). Mode of action: G. Leblondel, P. Allain, Biochem. Pharmacol. 27, 2099 (1978); J. R. O'Brien et al., Thromb. Res. 13, 245 (1978); J. J. Bruno, ibid. 1983, Suppl. 4, 59. Pharmacology: A. Akashi et al., Arzneim.-Forsch. 30, 409, 415 (1980). Clinical studies: J. J. Thebault et al., J. Int. Med. Res. 5, 405 (1977); C. Lecrubier et al., Therapie 32, 189 (1977); T. Katsumura et al., Angiology 33, 357 (1982). Review of pharmacodynamics, pharmacokinetics and therapeutic use: E. Saltiel, A. Ward, Drugs 34, 222-262 (1987). Comprehensive description: F. J. Al-Shammary, N. A. A. Mian, Anal. Profiles Drug Subs. Excip. 21, 573-609 (1992).
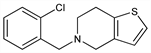
Derivative
Hydrochloride.Nomenclature
Properties
Crystals from ethanol, mp 190°. uv max (water): 214, 268, 295 nm (A1%1cm 303.8, 13.14, 2). pKa 7.64. Almost sol in water; sol in 95% alcohol, methanol, chloroform. Insol in ether. LD50 in mice (mg/kg/24 hrs): 55 i.v.; >300 orally (Castaigne).Therapeutic Category
Antithrombotic.
Keywords
Antithrombotic