9437. Tilidine
Nomenclature
CAS number: 20380-58-9
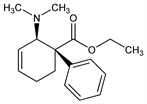
(1R,2S)-rel-2-(Dimethylamino)-1-phenyl-3-cyclohexene-1-carboxylic
acid ethyl ester; ethyl 2-(dimethylamino)-1-phenyl-3-cyclohexene-1-carboxylate; 3-trans-dimethylamino-4-phenyl-4-trans-carbethoxy-Δ1-cyclohexene. C17H23NO2; mol wt 273.37.
C 74.69%, H 8.48%, N 5.12%, O 11.71%.
Description and references
Opioid analgesic. Prepn: ZA 6606476; GB 1120186; G. Satzinger, US 3557127 (1967, 1968, 1971, all to Warner-Lambert). Synthesis of 6-C14 hydrochloride: K.-O. Vollmer, F. W. Koss, Arzneim.-Forsch. 20, 990 (1970). Pharmacology and toxicity data: M. Hermann et al., ibid. 977, 983. Metabolism: K.-O. Vollmer, H. Achenbach, ibid. 24, 1237 (1974). HPLC determn in pharmaceutics: D. Zivanov-Stakic et al., Farmaco 44, 759 (1989). Clinical trial in comparison with nefopam, q.v.: J. Abeloos et al., Acta Anaesthesiol. Belg. 34, 283 (1983); in combination with naloxone, q.v.: H. Van Cauwenberge et al., Int. J. Clin. Pharmacol. Res. 12, 1 (1992).
Properties
bp0.01 95.5-96°.Derivative
Hydrochloride hemihydrate.Nomenclature
G-1261C; W-5759A; Lucayan (Corvi); Valoron (Gdecke).C17H23NO2.HCl.H2O; mol wt 318.84.
C 64.04%, H 7.90%, N 4.39%, O 12.55%, Cl 11.12%.
Properties
Crystals, mp 125°. Easily sol in water. LD50 (7 day) in mice, rats (mg/kg): 437.0, 417.7 i.g.; 490.0, 400.0 s.c.; 52.0, 74.1 i.v. (Herrmann).Derivative
Hydrochloride.Nomenclature
CAS number: 27107-79-5
Properties
Crystals from ethyl acetate-methyl ethyl ketone, mp 159°.Derivative
cis-(±)-Form.Nomenclature
CAS number: 112244-09-4
Properties
bp0.01 97.5-98°.Derivative
cis-(±)-Hydrochloride.C17H23NO2.HCl.1H2O; mol wt 318.84.
C 64.04%, H 7.90%, N 4.39%, O 12.55%, Cl 11.12%.
Properties
Crystals from ethyl acetate-methyl ethyl ketone, mp 84°.Note
This is a controlled substance (opiate): 21 CFR, 1308.11.Therapeutic Category
Analgesic.
Keywords
Analgesic; Opioids