9444. Timolol
Nomenclature
Description and references
β-Adrenergic blocker. Prepn: B. K. Wasson, DE 1925956; idem, US 3655663 (1969, 1972 both to Frosst). Manufacturing process: L. M. Weinstock et al., DE 1925955; eidem, US 3657237 (1970, 1972 both to Frosst). Synthesis and activity data: Wasson et al., J. Med. Chem. 15, 651 (1972). HPLC determn in plasma: H. He et al., J. Chromatogr. B 661, 351 (1994). Pharmacology: Franciosa et al., Clin. Pharmacol. Ther. 13, 138 (1972); Ulrych et al., ibid. 232. Review of efficacy in glaucoma: R. C. Heel et al., Drugs 17, 38-55 (1979). Clinical evaluation in hypertension: B. A. Rofman et al., Hypertension 2, 643 (1980). Multicenter study of effect in myocardial infarction: N. Engl. J. Med. 304, 801 (1981); of efficacy in limiting infarct size: M. Sederholm et al., ibid. 310, 9 (1984). Comprehensive description: D. J. Mazzo, A. E. Loper, Anal. Profiles Drug Subs. 16, 641-692 (1987). Review of clinical experience with dorzolamide, q.v. in glaucoma and ocular hypertension: J. E. Frampton, C. M. Perry, Drugs Aging 23, 977 (2006).
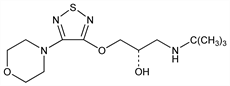
Derivative
Hemihydrate.Properties
White, odorless, crystalline powder. Slightly sol in water; freely sol in ethanol.Derivative
Hydrogen maleate salt.Nomenclature
Properties
White crystals from ethanol, mp 201.5-202.5°. [α]24405 12.0° (c = 5 in 1N HCl), [α]D25 4.2°. uv max (0.1N HCl): 294 nm (A1%1cm 200). Sol in water, ethanol, methanol; sparingly sol in chloroform; very slightly sol in cyclohexane. Practically insol in isooctane, ether. Stable in soln up to pH 12.Derivative
(±)-Free base.Properties
Crystalline solid from isopropyl ether, mp 71.5-72.5°.Therapeutic Category
Antihypertensive; antiarrhythmic (class II); antianginal; antiglaucoma agent.
Keywords
β-Adrenergic Blocker; Antianginal; Antiarrhythmic; Antiglaucoma; Antihypertensive; Aryloxypropanolamine Derivatives