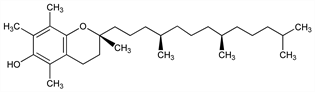
9495. α-Tocopherol
Nomenclature
Description and references
Most bioactive of the naturally occurring forms of vitamin E, q.v. Richest sources are green vegetables, grains, and oils, particularly palm, safflower and sunflower oils. Isoln from wheat germ: H. M. Evans et al., J. Biol. Chem. 113, 319 (1936). Structure: E. Fernholz, J. Am. Chem. Soc. 59, 1154 (1937); 60, 700 (1938). Synthesis of dl-form: P. Karrer et al., Helv. Chim. Acta 21, 520, 820 (1938); F. Bergel et al., J. Chem. Soc. 1938, 1382. Distillation from vegetable oils and prepn of esters: J. G. Baxter et al., J. Am. Chem. Soc. 918 (1943). Prepn of crystalline natural form: C. D. Robeson, ibid. 1660; of crystalline acetate: idem, ibid. 64, 1487 (1942). Abs config of natural α-tocopherol: H. Mayer et al., Helv. Chim. Acta 46, 963 (1963). Stereoselective synthesis: K.-K. Chan et al., J. Org. Chem. 43, 3435 (1978). Total synthesis of all 8 stereoisomers: N. Cohen et al., Helv. Chim. Acta 64, 1158 (1981). Clinical trial in Alzheimer's disease: M. Sano et al., N. Engl. J. Med. 336, 1216 (1997); to improve immune function in healthy elderly: S. N. Meydani et al., J. Am. Med. Assoc. 277, 1380 (1997). Review of bioavailability from vitamin E supplements: M. G. Traber, BioFactors 10, 115-120 (1999). Review of clinical trials in heart disease: W. A. Pryor, Free Radical Biol. Med. 28, 141-164 (2000).
Properties
Transparent needles, mp 2.5-3.5°. [α]25546.1 3.0° (benzene); [α]25546.1 +0.32° (ethanol).Derivative
Acetate.Nomenclature
Properties
Light yellow oil. Crystallized at 30° as needle-like crystals, mp 26.5-27.5°. [α]D25 +0.25° (c = 10 in chloroform); [α]D25 +3.2° (in ethanol).Derivative
Succinate.Nomenclature
Properties
Needles from petr ether, mp 76-77°. uv max (ethanol): 286 nm (E1%1cm 38.5). Practically insol in water.Derivative
dl-α-Tocopherol.Nomenclature
Description and references
Equimolar mixture of all four racemates.
Properties
Slightly viscous, pale yellow oil. d425 0.950; bp0.1 200-220°; nD25 1.5045. uv max: 294 nm (E1%1cm 71). Practically insol in water. Freely sol in oils, fats, acetone, alcohol, chloroform, ether, other fat solvents. Stable to heat and alkalies in the absence of oxygen. Not affected by acids up to 100°. Slowly oxidized by atm oxygen, rapidly by ferric and silver salts. Gradually darkens on exposure to light.Derivative
dl-α-Tocopherol acetate.Nomenclature
Description and references
Comprehensive description: B. C. Rudy, B. Z. Senkowski, Anal. Profiles Drug Subs. 3, 111-126 (1974).
Properties
Pale yellow, viscous liquid. mp 27.5°. d421.3 0.9533. bp0.01 184°; bp0.025 194°; bp0.3 224°. nD20 1.4950-1.4972. uv max (cyclohexane): 285.5 nm. Practically insol in water. Freely sol in acetone, chloroform, ether. Less readily sol in alc.Use
As an antioxidant in vegetable oils and shortening.Therapeutic Category
Vitamin E supplement.
Therapeutic Category (Veterinary)
Vitamin E supplement.Keywords
Vitamin/Vitamin Source; Vitamin E